Sponsored by :
OTT166 is an integrin inhibitor with anti-VEGF effects that shuts down negative signaling pathways in DR while activating the protective Tie2 pathway.
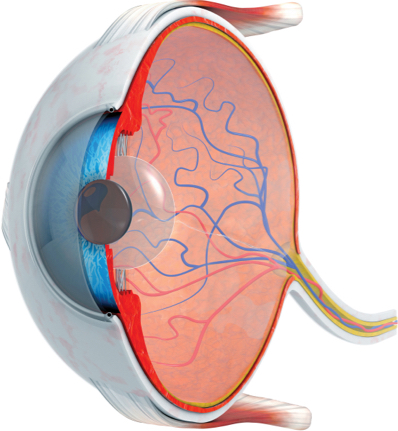
Purpose-engineered for topical administration, OTT166 is proven to diffuse to the retina via transscleral distribution and exhibit its highest concentration in the sclera, choroid, and retina (vs vitreous).
Preclinical studies and animal models show that OTT166 achieves therapeutic concentration ≥12 hours after dosing, with potent anti-VEGF activity comparable to intravitreal injection.
A phase 1 study in DR patients with DME showed overall safety, tolerability, and biological activity after 28 days of dosing. A phase 2 study in DR recently completed enrollment, with data expected to be reported in early 2024.
– Peter K. Kaiser, MD
Treatment of the patient with nonproliferative diabetic retinopathy (NPDR) is complicated. While anti-VEGF therapy has proven successful at reducing the risk of progression and in yielding regression in eyes with DR without center-involving diabetic macular edema (DME), the data to date show no benefit for visual outcomes.1 Moreover, starting a patient on treatment then obligates that individual to routine monitoring, follow-up, and continued intravitreal injections. For these reasons, most eyes with NPDR are currently followed without active therapy, and treatment is initiated upon signs of progression with loss of vision—admittedly an imperfect solution given the risk of losing a patient to follow-up.
But there is certainly good news for patients with NPDR. The innovation pipeline in DR is robust, to say the least, offering several candidate molecules acting on unique targets and featuring novel mechanisms of action and delivery. Altogether, and with the usual caveats about clinical development, clinicians can be optimistic that we are closing in on addressing a significant unmet need in DR.
I recently moderated a discussion panel on the current state of treatment in DR, where we focused on identifying current treatment gaps and looked down the developmental pipeline at some options that may soon be available for clinical use. There is one proposed molecule that seems to offer significant promise: OTT166 (OcuTerra) is an integrin inhibitor designed for topical delivery; after instillation onto the conjunctival surface, the small molecule travels to the retina transclerally and crosses the choroid, where it then affects multiple signaling pathways involved in DR pathophysiology including Tie-2, VEGF, PDGF, βFGF, and TGF-βR. If OTT166 is able to complete development and successfully navigate regulatory review, such a treatment would have tremendous impact as an easily used, patient-friendly option suitable for use in earlier stage disease. Having a topical therapy like this would propel a paradigm shift in the treatment of DR, allowing clinicians to be proactive instead of waiting for the disease to progress before intervening.
—Peter K. Kaiser, MD
Peter K. Kaiser, MD: Studies have shown that intravitreal anti-VEGF injections for the treatment of nonproliferative diabetic retinopathy (NPDR) in the absence of diabetic macular edema (DME) is associated with reduced diabetic retinopathy (DR) progression, 2-step or greater reduction in Diabetic Retinopathy Severity Scores (DRSS), and delay in onset of vision threatening complications, such as DME. However, according to the 2021 ASRS practice pattern, about 60% of retina specialists, both based in the US and internationally, said they closely monitor patients with NPDR off treatment. Overall, relatively few practitioners even considered offering anti-VEGF in a broad array of clinical scenarios.2 What is your take on why we see this divide between clinical trials and real-world practice?
David S. Boyer, MD: There could be several explanations. First, NPDR patients typically have good vision, so it may be difficult to motivate someone who is not experiencing any symptoms to start injections. Second, patients are anxious about starting injections and especially so when they are not convinced they have active disease. Third, as the PANORAMA and Protocol W studies showed, there was no significant visual improvement noted in the group treated with intravitreal anti-VEGF injections versus the untreated group.1-3 Fourth, the treatment burden adds to whatever other medical interactions these patients have due to their diabetes.
Dr. Kaiser: Our patients are stuck between a rock and a hard place: active monitoring is the preferred option for the majority of NPDR patients because active treatment engenders many questions.
Caroline R. Baumal, MD: While watchful waiting is a common paradigm, there are some key learnings about the natural history of severe NPDR from the PANORAMA study to be considered. Just over 50% of sham-treated (observed) eyes developed vision-threatening complications or center-involving DME by 100 weeks, which closely replicates findings from the ETDRS studies that were performed over 3 decades ago.4 It is telling that the rate of progression from severe NPDR to PDR with vision-threatening complications has not improved with better systemic medical therapies for diabetes.
There may be some scenarios when offering anti-VEGF treatment for severe NPDR without DME may be considered, for example, in a patient who had complications in the fellow eye related to PDR. It may also be considered in a patient with clinical signs of rapidly progressing NPDR severity and poor systemic control of diabetes. Anti-VEGF injections may be preferred over panretinal laser treatment for PDR in patients who cannot tolerate or cooperate with the laser procedure, in patients who have lost peripheral vision in one eye from laser, or in patients with a high risk of developing DME. I sometimes use fluorescein angiography findings to aid in the decision-making process. Signs of extensive vascular wall staining or leakage and/or retinal ischemia may prompt me to offer earlier treatment due to the high risk for progression.
+ Keep Reading
Dr. Kaiser: In patients for whom you are considering anti-VEGF injections, when do you start? How do you gauge the potential for adherence to the treatment regimen?
Dr. Boyer: The level of systemic glycemic control provides some information. An A1C hemoglobin of 9 or greater has me concerned about the patient’s compliance, and I would hesitate to start anti-VEGF injections alone. If the A1C is controlled, and if we determine that anti-VEGF is likely to have a benefit, then we explain that our objective is to prevent severe visual complications rather than to improve vision. We try to limit the number of injections needed to start, and we spread it out to eventually every 4 months.
The issue of adherence and compliance touches on a reason why there is so much optimism for treatments for NPDR that don’t require patients to come to the office. Something that could be prescribed for at-home use would facilitate earlier treatment, and the earlier we can treat, the higher the likelihood maintaining vision without complications.
Dr. Baumal: Monitoring adherence to treatment may benefit from advances in ophthalmic imaging. Recent advances in telemedicine, fundus photography, and at-home OCT suggest we may have an entirely new way of actively treating DR instead of monitoring disease.
+ Keep Reading
Dr. Kaiser: Is it possible that at least some of the suboptimal response to anti-VEGF injections that we observe is because pathways other than VEGF also drive the disease process?
Dr. Boyer: The answer, in a word, is yes. The DRCR.net Protocol I was a multicenter randomized clinical trial that compared intravitreal sham injection + prompt laser vs. intravitreal ranibizumab 0.5 mg + prompt laser vs. intravitreal ranibizumab 0.5.mg + deferred (after 6 months) laser vs. intravitreal triamcinolone 4 mg + prompt laser for diabetic macular edema. In the study, 40% of patients gained up to 12 letters of vision, and another 30% only gained around 6 letters.5 Additionally, some patients experienced no significant visual improvement. Taken together, the evidence suggests other mechanisms are involved, and that DR is a multifactorial disease. In DR and other retinal disorders, including neovascular age-related macular degeneration (AMD), the family of RGD-binding (arginine, glycine and aspartate) integrins amplify intracellular growth factor signaling, including VEGF, PDGF, βFGF, and TGF-βR, and offer another mechanism of action to treat patients.6
Dr. Kaiser: The DR pipeline is very active, with a wide gamut of programs looking at novel targets and delivery systems, including several focusing on this idea of affecting multiple signaling pathways.
Notably, the pipeline contains programs aiming for novel targets, as well as some going after novel mechanisms of delivery, including topical delivery. We do have to acknowledge that the topical approaches we are tracking today are unlike previous experiences with topical agents that simply placed an active into solution with no thoughts on whether the drug would make it to the posterior segment. We are now seeing topical approaches that have been specifically designed to target the posterior segment. One program is an eyedrop that diffuses to the retina via the transscleral route, delivering an integrin inhibitor that targets vitronectin and other ligands (OTT166). Integrins have attracted significant interest because of their role in regulating cell-cell and cell-extracellular matrix interactions. And so what is unique about this molecule is that it offers a novel mechanism of delivery (topical), aims at a novel target, and aims at multiple signaling pathways involved in driving DR pathology.
Dr. Boyer: OTT166 affects multiple integrins, including avβ3, avβ6, and avβ8, which are known drivers of inflammation, angiogenesis, fibrosis, and leakage, which combine to contribute to DR pathophysiology.7 Targeting multiple pathways has the potential to stop the source of this growth factor signaling that amplifies multiple signaling pathways: VEGF, PDGF, βFGF, and TGF-βR.7 As well, there is some suggestion that OTT166 upregulates the Tie-2 pathway, which we know is beneficial in DR and reduces retinal edema.
+ Keep Reading
► On Market
► Phase 1 - DR
► Phase 1 - DME
► Phase 2 - DR
► Phase 2 - DME
► Phase 3 - DME
► On Market
► Phase 1 - DR
► Phase 1 - DME
► Phase 2 - DR
► Phase 2 - DME
► Phase 3 - DME
1
2
► OTT166 (OcuTerra) – integrin inhibitor
► OCS-01 (Oculis) – steroid nanoparticle
► EXN-407 (Janssen) – SRPK-1 inhibitor
► PAN-90806 (PanOptica) – TKI inhibitor
► SYL-1801 (Sylentis) – RNA interference
► Eylea (aflibercept, Regeneron) – anti-VEGF
► Lucentis (Genentech) – anti-VEGF
► Vabysmo (Genentech) – anti-VEGF and Tie2 activator
► THR-149 (Oxurion) – plasma kallikren inhibitor
► AXT-107 (gersizangitide, AslepX) – anti-VEGF and Tie2 activator
► OTX-TKI (Ocular Therapeutics) – tyrosine kinase inhibitor
► Vorolanib (Eyepoint) – tyrosine kinase inhibitor
► PER-001 (Perfuse) – endothelin receptor agonist
► 4D-150 (4DMT) – intravitreal delivery
► RGX-314 (RegenXBio) – subcutaneous delivery
► APX-3330 (Ocuphire) – Ref-1 inhibitor
► OPL-0401 (Valo) – ROCK inhibitor
► MS-553 (MingSight) – PKC-Beta inhibitor
► RX-402 (Rezolute) – PKI
► HCB-1019 (InflammX) – Connexin43
► Runcaciguat (Bayer) - soluble guanylate cyclase
Purpose engineering means many things for OTT166, most notably in its mechanism of delivery. The molecule has been engineered with fluorine substitutions of hydrogen atoms in the molecule’s structure to increase membrane permeability.7 Following instillation to the ocular surface, OTT166 diffuses via the trans-scleral route through the conjunctiva, to the suprachoroidal space, to the choroid, and eventually to the retina, thus affecting diffusion via a concentration gradient from the outside of the globe. At the cellular level, OTT166 diffuses through the choroid and retina to exhibit powerful anti-VEGF effect, comparable to intravitreal injection.7,8 An additional novel aspect of OTT166 is its mechanism of action. OTT166 targets integrins, transmembrane proteins that bind with various ligands to initiate and accelerate multiple signaling pathways in DR (TGF-betaR; VEGF-A, -C, &-D; bFGF) associated with pathologic changes, including angiogenesis, exudation, inflammation, and fibrosis.6
Dr. Kaiser: The idea of using a drop to treat NPDR is very reasonable given that we are watchfully waiting currently. What is the evidence that OTT166 successfully reaches the retina to have an effect?
Dr. Boyer: The proof-of-concept evidence comes from a rabbit model in which therapeutic levels of OTT166 after topical administration were found to be at the highest concentration levels in the sclera and the retina/choroid compared to vitreous, aqueous, and plasma, thus supporting transscleral distribution.7 You can also find supporting evidence from early in-human trials in eyes with DME and AMD.8 The phase 1B studies were designed to look at safety, which was demonstrated: there were no drug-related serious adverse events or signs of corneal toxicity, other than one possibly drug-related case of a mild red eye. There was also a strong signal of biological activity, in terms of anatomic improvement of baseline DME.
Observations From Phase 2 of the DR:EAM Study
Carl D. Regillo, MD, FACS, discusses some observations from phase 2 of the DR:EAM study to evaluate the efficacy and safety of OTT166 ophthalmic solution (OcuTerra) to treat diabetic retinopathy. He comments on data from the phase 1 trial and explains how patients have responded during enrollment in phase 2.
Jeffrey S. Heier, MD: I was an investigator in the early-phase study. It’s difficult to extrapolate outcomes to eyes with NPDR, but there are some lessons we can learn. For instance, visual improvement, albeit minimal, was encouraging. To me, the anatomic improvement, demonstrated as resolution of fluid on OCT, was highly significant and strongly suggested that the topical application of OTT166 actually reached the retina, and then it had a positive effect in many patients once it got there (Figure 1).
Figure 1. A representative patient in the phase 1B study acheived improvement in DRSS score as early as day 28 after treatment.
The second takeaway is that there was a subgroup of patients who demonstrated a delayed response to treatment. At day 56, among responders, we observed greater reduction in mean retinal thickness compared to day 28, when treatment was stopped, which suggests durability of effect. Among previous nonresponders, there was a reduction in median retinal thickness, which suggests a delayed response in some eyes.
Dr. Boyer: A similar phenomenon has been demonstrated with previous intravitreal integrin development programs. What impressed me with OTT166 was that we were able to see a response at day 28 that in some patients continues through at least day 56. When we think about treating DME with anti-VEGF, we don’t always see a response that soon. As well, the safety associated with the higher dose fully justified further development.
Dr. Heier: The potential to have that kind of impact right away in early-stage DR seems incredibly promising. This drop would be something I could easily see myself considering for a wide range of NPDR patients, and possibly even some early DME eyes, where we can add anti-VEGF injections later if needed. There is potential for OTT166 to have an impact on slowing the progression to vision-threatening complications, because OTT166 has activity beyond the VEGF pathway.
Dr. Baumal: Of note, the phase 1B study enrolled a diverse population: this was an all-comers analysis, and some of these eyes had received numerous previous anti-VEGF injections. Yet, the results showed resolution of retinal thickness in this difficult-to-treat population by days 28 and 56. This is a rather impressive display of biologic plausibility in a challenging cohort.
Dr. Kaiser: The results of this study have shown evidence of treatment effect and a rapid response among responders. Again, we wouldn’t expect to see improvement in DRSS scores by 1 month after treatment with anti-VEGF injections, but that was seen in some eyes in the phase 1B study with the OTT166 eye drop.
+ Keep Reading
For more discussion and additional perspectives about cases from the phase 1b study, vist the OcuTerra Spotlight on Eyetube.
While developing OTT166, OcuTerra has engaged with the retina community to share its story and to listen for feedback. In those interactions, the company has heard some repeated questions about what, exactly, integrins do, and about whether a topical drop can reach the back of the eye. As well, the fact that the mechanism of delivery is a drop, and therefore depends on patients’ regular use, has raised questions about potential compliance. The last one may be the most complicated to answer, especially with respect to a molecule still in clinical trials. I will say that in my experience as the principal investigator for the phase 2 study, getting patients to buy in on the concept of topical therapy was easy. In my clinical practice, I often hear patients ask whether there is an alternate to anti-VEGF injections, a drop perhaps. Taken together, there is at least anecdotal evidence that patients might be accepting of a treatment paradigm that includes topical therapy.
I sat down with two colleagues, David Eichenbaum, MD, and Anat Loewenstein, MD, to discuss some of the feedback OcuTerra has received. Watch the full discussion in video format on Eyetube.
— Carl D. Regillo, MD
Read below for common myths the panel discussed and click to reveal the facts:
A topical formulation can’t reach the back of the eye
see the facts
OTT166 IS purpose-engineered for transscleral delivery
• OTT166 is applied to the conjunctiva, then diffuses through the sclera,9 permeates the choroid-RPE,10 and eventually diffuses through all retinal layers
• In a rabbit model, OTT166 concentration was found highest in the sclera, followed by the retina-choroid-plexus—and in both cases, exceeding the therapeutic concentration at least 12 hours after dosing
• Concentration was lowest in the plasma, followed by the aqueous humor and vitreous7
• In the same study, a 5% dose delivered QD was found to exhibit equivalent inhibition of vascular leakage as anti-VEGF injections7
• Taken together, the evidence supports trans-scleral delivery and diffusion (ie, concentration highest from the outside in), has a therapeutic effect at the tissue of interest, and demonstrates anti-VEGF activity
Integrin inhibition is not viable nor proven
see the facts
FACT: There is growing evidence across medicine supporting integrin inhibition, and there is evidence indicating biologic activity with OTT166 in human studies8
• An extensive library supporting integrin inhibition exists in the systemic literature, with applications in oncology, cardiology, rheumatology, and dermatology
• The parent molecule of OTT166 was proven effective in treating inflammation associated with rheumatoid arthritis
• Lifitegrast ophthalmic solution 5% (Xiidra, Novartis) is an approved integrin inhibitor in ophthalmology
• OTT166 was evaluated for safety and indication of biologic activity in a phase 1b study of patients with diabetic macular edema8
• Patients were followed for 28 days on treatment, then 28 days off
• Among responders (defined per protocol), mean central retinal thickness (CRT) decreased significantly on day 28, and mean CRT decreased again by day 56
• Even among nonresponders, the median CRT decreased from day 28 to day 56, suggesting a late-response
• 44% of subjects with interpretable color fundus photos showed a reduction in DR severity with just 28 days of treatment
Compliance will necessarily be an issue with topical therapy
see the facts
FACT: Maybe not!
• Patients’ education and how the concept of topical therapy is introduced in the clinic will be an important determinant of success
• It may be difficult to convince asymptomatic patients to accept an intervention, but if they are reminded that the alternate is to receive an injection in the eye, they might be inclined toward the less-invasive option—could this be a potential positive for compliance?
• There is a net benefit of having an option that addresses a current unmet need in DR treatment
• Especially because earlier intervention may forestall development of vision-threatening complications
• A topical therapy empowers patients to participate in their own treatment, which again may be motivating factor in inspiring compliance
Fact or Fiction: Can a Topical Formulation Reach the Retina?
The panel discusses feedback that OcuTerra has received about topical therapies it is developing for the treatment of diabetic retinopathy. They explain how preclinical and phase 1b data show efficacy for transmembrane permeability through the sclera.
Fact or Fiction: Are Integrin Inhibitors a Viable Strategy?
The panel discusses what the literature shows regarding the use of integrin inhibitors to treat systemic diseases. They also explain the growing body of evidence in phase 1 data for the efficacy of OTT166 for treating diabetic macular edema, and comment on a phase 2 trial to further examine its efficacy for treating diabetic retinopathy.
Fact or Fiction: Will Compliance Be an Issue With a Topical Drop for DR?
The panel dispel myths surrounding compliance with topical treatments for diabetic retinopathy. Dr. Loewenstein shares how she educates patients about using a less invasive topical drug. Dr. Eichenbaum highlights how such medications can help reduce the burden of the disease and delay progression with safe and effective treatment options.
Dr. Kaiser: A phase 2 study, the DR.EAM Study, recently completed enrollment, with an expected data readout in early 2024. What is important to know about this study?
Dr. Boyer: The phase 2 study is a multicenter, randomized, double-masked, placebo-controlled trial in eyes with moderately severe to severe NPDR to mild PDR, and no center-involved DME. The primary endpoint is a 2-step or greater improvement in DRSS at 24 weeks. The study will also look at some key secondary endpoints, including the prevention of progression to sight-threatening complications, delaying the time to intravitreal injection and/or PRP, and some exploratory imaging endpoints that could help shape our understanding of DR.
Dr. Baumal: The concept of a topically applied treatment that prevents DR progression and may yield regression of DRSS in eyes with NPDR warrants interest. We hear often about potential paradigm shifts, and they do not always pan out. With OTT166, the early data are very promising. The phase 2 results will tell us more of the story. If this molecule can be successfully developed, we would have a treatment that represents a huge improvement over the current preferred practice of watchful waiting of severe NPDR. We could treat patients proactively instead of passively monitoring them.
Dr. Heier: There is clinical potential beyond DR, as well. In the phase 1, there were some promising outcomes in eyes with AMD. Whether this is a safe and effective monotherapy or adjunctive therapy in AMD is a question for later down the line, but the concept of targeting multiple pathways makes sense. We’re already seeing the proof-of-concept in that with Ang2-targeting agents.
Dr. Kaiser: As with all development programs, cautious optimism is warranted. The potential to offer patients with NPDR an easy self-administered, topical therapy that could reduce DR progression, or even yield regression and delay or even eliminate the need for invasive injections, lasers, or both, would transform DR management. We have evidence that OTT166 reaches the retina in therapeutic levels, early confirmation of benefit from a phase 1 study, and now we await results from a crucial phase 2 study. If the data come back positive, we are on the precipice of a true paradigm shift in decision-making for patients with early-stage DR: we could potentially be entering an era of active, noninvasive, and proactive treatment, instead of monitoring as a default because of the myriad issues with current treatment options.
References
1. Brown DM, Wykoff CC, Boyer D, et al. Evaluation of intravitreal aflibercept for the treatment of severe nonproliferative diabetic retinopathy: results from the PANORAMA Randomized Clinical Trial. JAMA Ophthalmol. 2021;139(9):946-955.
2. Hahn P, ed. ASRS Preferences and Trends Membership Survey. Presented at: American Society of Retina Specialists. Oct. 8-12, 2021; Chicago.
3. Maturi RK, Glassman AR, Josic K, et al; DRCR Retina Network. Four-year visual outcomes in the protocol w randomized trial of intravitreous aflibercept for prevention of vision-threatening complications of diabetic retinopathy. JAMA. 2023;329(5):376-385. Erratum in: JAMA. 2023;329(12):1034.
4. Early Treatment Diabetic Retinopathy Study Research Group. Fundus photographic risk factors for progression of diabetic retinopathy. ETDRS report number 12. Ophthalmology. 1991;98(5 Suppl):823-833.
5. Bressler SB, Qin H, Beck RW, et al; Diabetic Retinopathy Clinical Research Network. Factors associated with changes in visual acuity and OCT thickness at 1 year after treatment for diabetic macular edema with ranibizumab. Arch Ophthalmol. 2012;30:1153-1161.
6. Van Hove I, Hu TT, Beets K, et al. Targeting RGD-binding integrins as an integrative therapy for diabetic retinopathy and neovascu-lar age-related macular degeneration. Prog Retin Eye Res. 2021;85:100966.
7. Askew BC, Furuya T, Edwards DS. Ocular distribution and pharmacodynamics of SF0166, a topically administered avβ3 integrin antagonist, for the treatment of retinal diseases. J Pharmacol Exp Ther. 2018;366(2):244-250.
8. Boyer DS, Kaiser PK, Magrath GN, et al. The safety and biological activity of OTT166, a novel topical selective integrin inhibitor for the treatment of diabetic eye disease: a phase 1b study. Ophthalmic Surg Lasers Imaging Retina 2022;53:553-560.
9. Loch C, Zakelj S, Kristl A, et al. Determination of permeability coefficients of ophthalmic drugs through different layers of porcine, rabbit and bovine eyes. Eur J Pharm Sci. 2012;47(1):131-138.
10. Pitkänen L, Ranta VP, Moilanen H, Urtti A. Permeability of retinal pigment epithelium: effects of permeant molecular weight and lipophilicity. Invest Ophthalmol Vis Sci. 2005;46(2):641-646.