Sponsored by ZEISS
Clínica de Olhos Leitão Guerra, Salvador, Brazil
Age-related macular degeneration (AMD) is the leading cause of severe, irreversible vision loss in people over age 60, and it is growing at an alarming rate all over the world. Your patients may be at risk. It is estimated that 196 million people had AMD in 2020, with that number rising to 288 million by 2040, yet 25% of AMD in the primary eyecare setting goes undiagnosed.1,2
By combining premier imaging and at-a-glance history with interactive analysis, ZEISS Retina Workflow solutions support both your routine and complex patient management decision processes with an efficient, worry-free and cost effective solution.
Join me as I demo the ZEISS Retina Workflow, using actual patient cases, to show how this solution helps improve efficiency in my practice and can also help you provide better care to your AMD patients.
1. Wong, Wan Ling, et al. "Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis." The Lancet Global Health 2.2 (2014): e106-e116
2. Neely DC, Bray KJ, Huisingh CE, et al. Prevalence of undiagnosed age-related macular degeneration in primary eye care. JAMA Ophthalmol. 2017;135(6):570-5
Ricardo Leitão Guerra, MD, MSc, FICO
See an overview of the ZEISS Retina Workflow.
No time for a video? Scroll down to see a series of animated GIFs featuring explanations from Dr. Guerra.
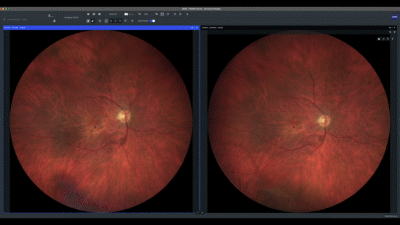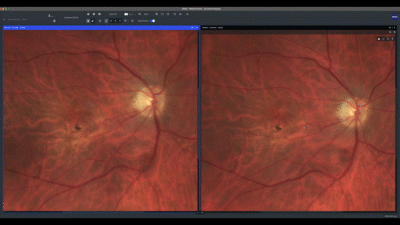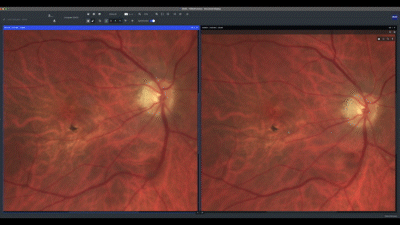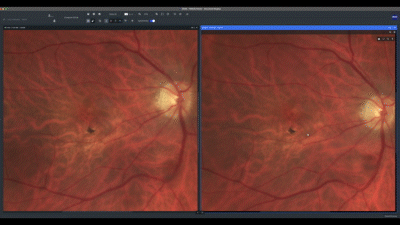
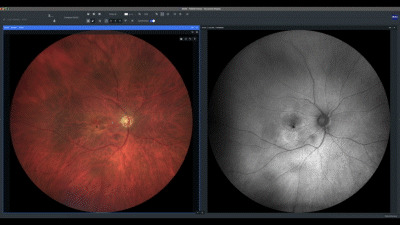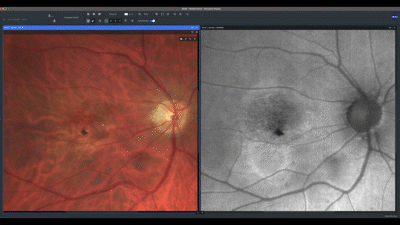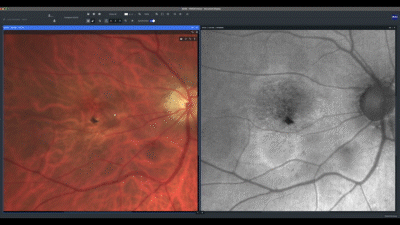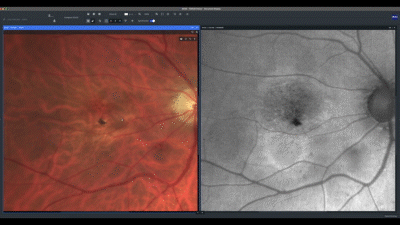
I always start by analyzing true color images from the ZEISS CLARUS 700, and love working with the synchronization mode activated on the ZEISS Retina Workplace application, which allows for the simultaneous manipulation of multiple images. The separation of the RGB color channels helps me identify abnormalities in different structures.
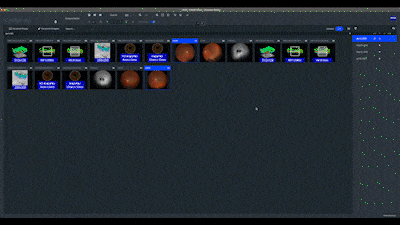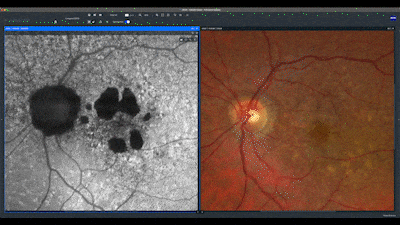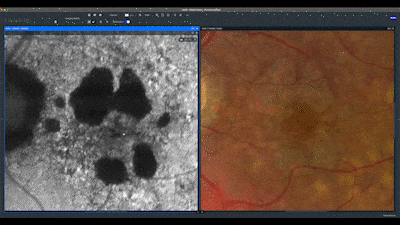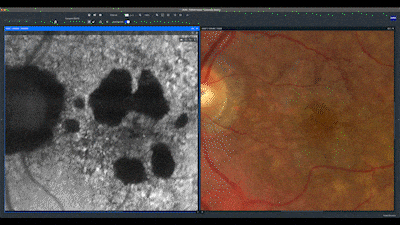
Next, I evaluate each eye individually, beginning with the correlation of the true color and fundus auto-fluorescence images. For AMD patients, I prefer using the green wavelength, which has less absorption by the macular yellow pigment. In this case, we can see an area of marked high auto-fluorescence corresponding to RPE atrophy.
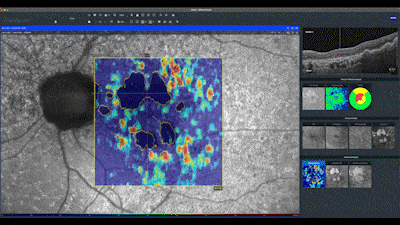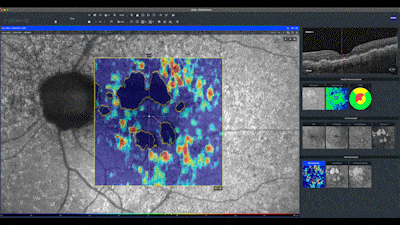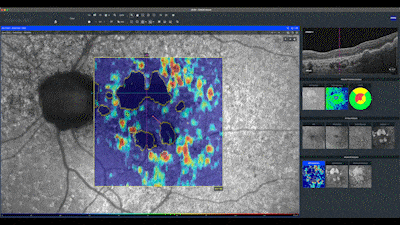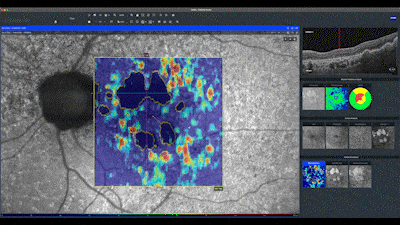
I advise using the qualitative and quantitative analysis of the ZEISS CIRRUS OCT, always starting with the unphased maps generated by the macular cube scan. For cases of AMD, I use customized segmentation for the qualitative analysis of the choroid's hyper-transmission defects, setting compensation at 64 and thickness at 400.
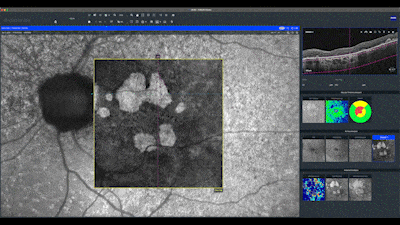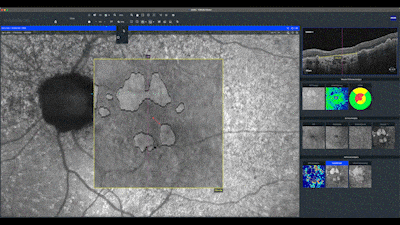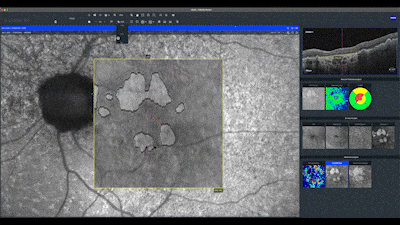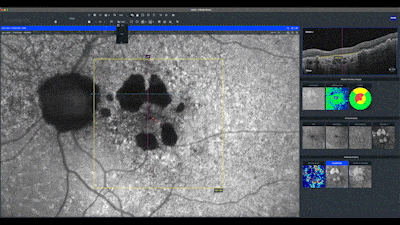
In this en face map, lighter areas correspond to RPE hyper-transmission defects and assist in identifying small defects not yet detected in the quantitative analysis of the sub-RPE slab map. Notice in this case how the automatic analysis precisely delineates the boundaries of the geographic atrophy seen as hypo-auto-fluorescent areas. The red line on this map indicates the distance of the atrophy from the center of fovea.
The advanced analysis map of the RPE, only available on the ZEISS CIRRUS OCT, presents this delineation of the atrophy by a yellow line, associated with the visualization of the RPE elevations in a color scale.
Wet AMD
Geographic Atrophy
Ricardo Leitão Guerra, MD, MSc, FICO
Dr. Guerra shares the case of a 77-year-old man with neovascular AMD OD and dry AMD OS. Join him as Dr. Guerra demonstrates how the ZEISS Retina Workplace helps him track the patient as he progresses from dry AMD to geographic atrophy.
No time for a video? Scroll down to see a series of animated GIFs featuring explanations from Dr. Guerra.
A true color image of the left eye at baseline shows confluent soft drusen and pigment mobilization. When comparing the baseline image with the current one, we notice a reduction in the number of drusen. The green channel aids in this visualization.
Quantitative analysis of the macular cube OCT scans shows the retina thickness temperature map presenting unremarkable changes.
Here, an RPE advanced analysis map highlights reabsorption of the drusen. Notice the obvious reduction in RPE elevations, represented by warm colors.
The graphic at the bottom of the screen helps us to intuitively understand this evolution, and the quantitative values can be accessed by clicking on the icon in the lower left table. Also note that the software can identify an area of RPE atrophy, demarcated by a yellow line.
When analyzing the image overlaid on the FAF exam, we notice the perfect correlation of the RPE atrophy area with hypoautofluorescence.
The en face image created by a customized choroid segmentation using compensation at 64 and thickness at 400 allows the identification of small hypertransmission defects at the baseline exam.
A barcode sign can be observed in the B scan. Both findings are biomarkers for progression to advanced forms of AMD.
In this case, at the final follow-up, this hyper-transmission defect progressed to a small area of complete atrophy of the RPE and outer retina.
Ricardo Leitão Guerra, MD, MSc, FICO
Dr. Guerra shares the case of an 80-year-old man who presented with neovascular AMD and a 5-year history of anti-VEGF treatment. See how Dr. Guerra uses the ZEISS Retina Workplace to evaluate this patient.
No time for a video? Scroll down to see a series of animated GIFs featuring explanations from Dr. Guerra.
I always start cases such as this one by comparing the current clinical image with a previous one. In this case, both images show the same aspects.
Next, I analyze fundus autofluorescence scans. In this case, we see a central hypo-autofluorescent area with a gravitational hyper-autofluorescence sign corresponding to the area affected by subretinal fluid.
Structural OCT evaluation is a key aspect of the Retina Workplace. It facilitates the simultaneous assessment of exams conducted at different times. The lower timeline bar is a visual representation of the patient's interventions, including anti-VEGF treatment. Note that different medications are denoted with distinct colors.
The en face maps are intuitive and effective for monitoring the progression in cases treated with anti-VEGF.
The minimum intensity map reveals dark areas indicative of fluid presence, which simplifies the analysis of the case's evolution. To differentiate between intraretinal and subretinal fluid, I employ mid and ellipsoid zone maps.
The drag-and-drop functionality of exams on the timeline streamlines and enhances the analysis.
Furthermore, an OCT angiography exam was performed to evaluate the biomarkers of the neovascular membrane. In cases of type 1 macular neovascularization, I use a customized map with specific segmentation and a modified sub-RPE map, which also utilizes a tailored segmentation approach for clearer visualization.
In this patient, the automatic segmentation was exceptionally accurate as confirmed by the b-scan analysis.
Ricardo Leitão Guerra, MD, MSc
The ZEISS Retina Workflow integrates seamlessly into my clinical routine. When examining patients with AMD who have been under my care for several visits, I can easily compare their most recent imaging results with data from months or years ago. If pathology were to be detected on a recent visit, I align their most recent photographs or images with corresponding photographs or images so I can track the progression of their disease AMD—and possibly anticipate any forthcoming pathology.
For new patients with AMD, the ZEISS Retina Workflow empowers me to start a patient's journey from a position of technologic strength. New patients have the unique privilege of unlocking the power of the latest imaging modalities and software frameworks, and they build a robust library of patient-specific data as their journey grows.
When examining patients with AMD who have been under my care for several visits, I can easily compare their most recent imaging results with data from months or years ago. … For new patients with AMD, the ZEISS Retina Workflow empowers me to start a patient’s journey from a position of technologic strength.
The statements of the doctors reflect only their personal opinions and experiences and do not necessarily reflect the opinion of any institution with whom they are affiliated. The doctors alone are responsible for the content of their experience reported and any potential resulting infringements. Carl Zeiss Meditec, Inc and its affiliates do not have clinical evidence supporting the opinions and statements of the doctors nor accept any responsibility or liability of the authors' content.
Products, services or offers referenced on this page may not be available in all countries and product labeling varies by country.
Not all products, uses, treatment options and protocols referenced are officially approved in every market or supported by a product’s intended use in every market. Approved labeling and instructions may vary from one country to another. For country-specific product information, see the appropriate country website. Product specifications are subject to change in design and scope of delivery as a result of ongoing technical development.
en-INT_31_174_0262II