

Traditional therapies for treatment of retinal diseases have centered around surgery, laser, and related approaches, but the past decade has seen a shift toward targeted molecular therapies that directly interfere with pathologic molecular pathways. Most notably, this approach has been fruitful for the treatment of retinal diseases mediated by VEGF, such as diabetic retinopathy, diabetic macular degeneration, and cytomegalovirus (CMV) retinitis.
AT A GLANCE
• Cellular therapy is the administration of intact, living cells to patients for the treatment of disease.
• Cellular immunotherapy harnesses the body’s immune system in either autologous or allogeneic fashion to induce a highly specific immune attack on target cells.
• Cellular immunotherapy for CMV retinitis with CMV-specific cytotoxic T-cells involves systemic, intravenous infusion of partially matched donor cytotoxic T-cells sensitized in vitro to the pp65 CMV antigen.
The growth of targeted therapies beyond those impinging upon the VEGF pathway has been slow, but a number of other targeted molecular therapies for retinal disease are in the research pipeline. These targeted molecular therapeutics are likely to continue to fuel tremendous advances in the field of retina; however, another approach to retinal therapeutics represents a new frontier: developing strategies that exploit the body’s own cellular machinery.
Gene therapy, in which genetic material is delivered intracellularly to replace a dysfunctional gene product or to generate a protein that directly targets a disease-related molecular pathway, is one such strategy.1 Cellular therapy, also called cell-based therapy, is another. This article provides an overview of cellular immunotherapy in the treatment of CMV retinitis.
CELLULAR IMMUNOTHERAPY 101
Cellular therapy is the administration of intact, living cells (eg, stem cells and mature cells) to patients to treat disease.
In stem cell therapy, unipotent or multipotent cells are differentiated into a specific cell type and integrated into a site of injury to replace damaged tissue, ultimately improving the function of the target tissue or organ. Stem cells could potentially be used to replace neural retinal cells (eg, photoreceptors) or supporting cells (eg, retinal pigment epithelium) lost in retinal diseases such as retinal degenerations.2
Mature cells have been used to treat disease through cellular immunotherapy, in which intact, fully differentiated immune cells are administered to patients. This approach has begun to revolutionize the treatment of certain cancers through the use of, for example, chimeric antigen receptor (CAR) T-cells to treat acute lymphoblastic leukemia. This form of cell transfer has been likened to giving patients a living drug.
How It Works
Immunotherapy harnesses the body’s immune system to combat disease. Cellular immunotherapy involves administration of immune cells in either an autologous or allogeneic fashion to induce a highly specific immune attack upon target cells. Other forms of immunotherapy include the use of cytokines, monoclonal antibodies, vaccines, and toll-like receptor agonists.
In autologous cellular therapy, the patient’s own immune cells are harvested, engineered and/or expanded ex vivo to target specific antigens, and then reintroduced into the patient.
In allogeneic cellular therapy, donor immune cells are used. The allogeneic approach offers the potential to administer unmatched or partially matched allogeneic cells in an off-the-shelf manner.
Initial Uses
Cellular immunotherapy has its roots in oncology, where a number of approaches have been employed, including adoptive cell therapy (ACT), which involves infusion of immune cells that specifically target tumor cells. In adoptive T-cell transfer therapy, immune cells can be infused several ways. Autologous T-cells with antitumor activity can be isolated from the patient’s tumor or peripheral blood and expanded to a large number of cells ex vivo. These T-cells can then be infused back into the patient to generate a robust antitumor immune response.
For example, tumor-infiltrating lymphocytes (TIL) found within a patient’s tumor can be selectively cultured from tumor biopsies and expanded to a large number ex vivo before being infused back into the patient.3,4 Much of the work in ACT using TILs has traditionally been in patients with highly advanced refractory metastatic melanomas, and in this setting these patients have demonstrated durable response rates of approximately 50% in multiple trials.3,5,6 TILs have recently been investigated in a number of other cancers.3
Alternatively, rather than selecting for T-cells that already have antitumor activity and then expanding them, autologous or donor T-cells from the peripheral blood can be sensitized or genetically engineered (through CAR T-cell receptor changes) in vitro to recognize tumor antigens.4 Similar ACTs have also been proposed using natural killer cells instead of T-cells.7
Dendritic cell−based immunotherapies, also known as cancer vaccines, involve administering immune cells to prime the immune system against tumor cells. Dendritic cells are harvested and either pulsed with antigens or genetically modified to activate them against a specific antigen. Once transfused into a patient, the activated dendritic cells present tumor antigens to effector lymphocytes, thereby inducing a specific immune response against the tumor cells bearing these antigens. This approach has been used with success in patients with metastatic prostate cancer.8
CELLULAR IMMUNOTHERAPY IN CMV RETINITIS
Recently, researchers have begun carving out a role for
a cell-based therapeutic approach in retinal disease—specifically, for CMV retinitis.
CMV is a member of the herpes viral family. Infection with CMV is common in the general population, but clinically significant infection typically occurs in immunocompromised patients (eg, those with HIV, those who have had stem cells transplanted, and those who have received solid organ transplants).
In CMV retinitis, it is thought that viral particles in the blood enter retinal vascular endothelial cells through endocytosis, resulting in disruption of the blood-retina barrier, and thereafter access the retinal tissues.9 CMV retinitis generally begins peripherally and spreads centrifugally, with profound visual loss occurring once the infection spreads to the macula or nerve, or when retinitis leads to a retinal detachment.10 CMV retinitis occurs in patients who are unable to generate a sufficient primary T-cell response against the virus.
Considerations in Immunocompromised Patients
The 4-year incidence of CMV retinitis in patients with HIV with a CD4 count of less than 100 cells/mL was 25% in the era before the development of highly active antiretroviral therapy (HAART).11 The widespread use of HAART has reduced the rate of CMV retinitis to 7%, although there remains a significant cohort of HIV patients who nevertheless progress to AIDS and are at risk for opportunistic infections including CMV retinitis.10,12 HIV patients most at risk for CMV retinitis are those who are noncompliant with, intolerant of, or unresponsive to HAART, although CMV retinitis can occur even in the setting of immune reconstitution on HAART with high CD4 count, low viral load, or both.13 Moreover, even in the era of HAART, it has been shown that CMV retinitis is a condition wrought with recurrences and risk of progression and/or vision loss, even in patients with immune reconstitution.14-16
In the HAART era, CMV retinitis is also increasingly encountered in transplant patients, especially those on potent immunosuppressive therapy. In patients who have received a solid organ transplant, the risk of CMV retinitis is highest when the recipient is CMV negative and the donor is CMV positive, as the transplanted organ introduces CMV into a recipient who lacks effective immunity to the virus. In patients who have received allogeneic bone marrow or stem cell transplants, the risk of CMV infection is particularly high when the recipient is CMV positive but the donor is CMV negative. This is because the chemotherapy eradicates not only the tumor cells but also the recipient’s own immune cells. Also, the CMV-negative donor’s immune cells are not sensitized to CMV, so the patient is particularly susceptible to CMV infection. The risk of CMV retinitis in these post-transplant patients is further potentiated by immunosuppressive medications for the treatment of the malignancy or treatment or prevention of graft rejection or graft-versus-host disease. More recently, cases of CMV retinitis have also been reported following intraocular or periocular steroid administration in immunocompetent patients.17
Treatment of CMV Retinitis
The current mainstay of CMV retinitis treatment is immune reconstitution plus systemic and/or intravitreal antiviral agents.
Immune Reconstitution
Immune reconstitution is the cornerstone of CMV retinitis treatment. Effective antiretroviral therapy (ART) in patients with HIV disease can durably suppress HIV replication and typically results in rapid and sustained rise in CD4 counts. The recovery of CD4 levels leads to decreased incidence of HIV-associated opportunistic infections, including improvements in immune response to CMV. This represents the immune reconstitution that is the foundation of CMV retinitis therapy. Unfortunately, immune reconstitution is not possible in certain patients, including those requiring chronic immunosuppression (eg, solid organ transplant patients and leukemic patients who are status post bone marrow stem cell transplant) and those whose HIV is resistant or refractory to ART.
Antiviral Agents
Antiviral medications for CMV include ganciclovir (Zirgan, Bausch + Lomb; Vitrasert, Auritec Pharmaceuticals; and Cytovene, Genentech), valganciclovir (Valcyte, Genentech), cidofovir (Vistide, Gilead Sciences), and foscarnet (Foscavir, Hospira). Ganciclovir, and its prodrug valganciclovir, are phosphorylated by the viral kinase UL97 and subsequently by host kinases to ganciclovir triphosphate, which inhibits the viral DNA polymerase UL54. Mutations in UL97 therefore confer resistance to ganciclovir and valganciclovir. Cidofovir is phosphorylated by host kinases to its active form, which also inhibits viral DNA polymerase UL54. Foscarnet directly inhibits the viral DNA polymerase UL54, and mutations in UL54 can cause broad resistance to ganciclovir, valgancyclovir, foscarnet, and cidofovir.18 Intravitreal ganciclovir injection may have efficacy in the retina even when the CMV is “resistant” to CMV due to UL97 or UL54 mutations, given the high concentrations achievable in the vitreous through local administration. Systemic side effects, including renal toxicity (foscarnet and cidofovir) and bone marrow suppression (ganciclovir and valgancyclovir), can also limit the use of these agents.
It is important to note that a given strain of CMV may possess mutations that confer resistance to antiviral therapy. Furthermore, the systemic side effects of the antiviral treatments themselves may limit their utility. Even patients in whom antiviral therapy can be administered and in whom immune reconstitution can be achieved are at risk of recurrences, progression, and/or vision loss from CMV retinitis, as well as second-eye involvement in patients presenting with unilateral disease.14-16 Moreover, since the manufacture of the ganciclovir intraocular implant was discontinued, treatment of CMV retinitis now often involves significant morbidity and inconvenience to patients, often requiring frequent intravitreal injections (sometimes with multiple antiviral agents) for an extended period of time.
CMV-Specific Cytotoxic T-Cells
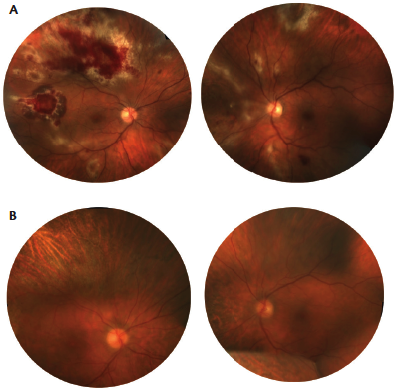
Figure. A chronically immunosuppressed patient status post allogeneic stem cell transplant on immunosuppression for severe graft-versus-host disease. Patient presented with resistant CMV viremia and active, vision-threatening CMV retinitis in both eyes. Color images of the right and left eyes show active CMV retinitis despite biweekly bilateral anti-CMV injections (A). Color images of the right and left eye more than 2 years after two cycles of systemic CMV specific T-cell infusions (B). The retinitis is completely regressed, as is the systemic CMV viremia with all systemic and intraocular CMV therapy ceased. There was no obvious worsening of the patient’s graft-versus-host disease nor any signs of ocular inflammation during or after systemic T-cell therapy.
Cellular immunotherapy with CMV-specific cytotoxic T-cells is another approach to treating CMV retinitis. It involves systemic, intravenous infusion of partially matched donor cytotoxic T-cells sensitized in vitro to the pp65 CMV antigen.19 A library of these cells was developed by a team at Memorial Sloan-Kettering Cancer Center (MSKCC),19 and the cells have already demonstrated efficacy against persistent CMV viremia or systemic infection in the stem cell transplant population.20
Recently we, in collaboration with MSKCC, have reported the application of this therapy specifically for patients with CMV retinitis who are resistant to, refractory to, or intolerant of systemic and/or intravitreal antiviral therapies.21 Under the leadership of our partners at MSKCC, we have developed a protocol to treat chronically immunosuppressed patients secondary to solid organ transplant (eg, kidney transplant, heart transplant), resistant HIV, or bone marrow transplant using systemically infused CMV specific T-cells (Figure).
Interestingly, even in the context of the blood-retina barrier, intravenously administered T-cells are able to target the CMV retinitis within the eye. Most often, patients in whom CMV is active in the eye have little or no signs of systemic CMV viremia. Therefore, we typically follow the response to the T-cell infusions with serial ophthalmic evaluations including dilated fundus photography, angiography, optical coherence tomography, and CMV polymerase chain reaction from successive anterior chamber taps. Most of our patients receive two or three rounds of T-cell infusions, each round consisting of three sessions of infusions, with several weeks between successive rounds of T-cells. In addition to the ophthalmic monitoring of response, systemic biomarkers (including cytokine profile and specific infused cell detection) are also used to gauge the level of response in each patient.
Although still preliminary, our initial results indicate that this is a viable strategy for the treatment of patients in whom there is no realistic alternative. In patients who have been treated thus far, sustained remission of CMV retinitis appears to be the general rule without any significant or severe local or systemic adverse events. Longer-term follow-up as well as use in a larger patient population will undoubtedly have to be closely evaluated before such therapy becomes more widespread and standard of care.
CONCLUSION
Given the limitations in the treatment of CMV retinitis, the field is ripe for novel treatment strategies. It seems that cellular immunotherapy with CMV-specific cytotoxic T-cells may be one such option. Further studies are necessary to more definitively parse out the role of adoptive immunotherapy, including its long-term efficacy and safety, for the treatment of CMV retinitis. Nevertheless, our early experience with this treatment approach is extremely encouraging and suggests a potential role for cellular immunotherapy in the treatment of retinal disease. n
Note: Should any reader have a potential patient who may benefit from this treatment, please contact Dr. Kiss directly.
1. Seidman C, Kiss S. Gene therapy: the next frontier for treatment of acquired and inherited ocular disorders. Retina Today. 2015;10(7):69-71.
2. Shapiro A. The future of stem cell therapy in retina. Retina Today. 2015;10(4):22-23.
3. Besser MJ, Shapira-Frommer R, Schachter J. Tumor-infiltrating lymphocytes: clinical experience. Cancer J. 2015;21(6):465-469.
4. Feldman SA, Assadipour Y, Kriley I, et al. Adoptive cell therapy-tumor-infiltrating lymphocytes, T-cell receptors, and chimeric antigen receptors. Semin Oncol. 2014;42(4):626-639.
5. Hong JJ, Rosenberg SA, Dudley ME, et al. Successful treatment of melanoma brain metastases with adoptive cell therapy. Clin Cancer Res. 2010;16(19):4892-4898.
6. Rosenberg SA, Yang JC, Sherry RM, et al. Durable complete responses in heavily pretreated patients with metastatic melanoma using T-cell transfer immunotherapy. Clin Cancer Res. 2011;17(13):4550-4557.
7. Eguizabal C, Zenarruzabeitia O, Monge J, et al. Natural killer cells for cancer immunotherapy: pluripotent stem-cell derived NK cells as an immunotherapeutic perspective. Front Immunol. 2014;5:439.
8. Kantoff PW, Higano CS, Shore ND, et al; for the IMPACT Study investigators. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Eng J Med. 2010;363(5):411-422.
9. Bodaghi B, Slobbe-van Drunen ME, Topilko A, et al. Entry of human cytomegalovirus into retinal pigment epithelial and endothelial cells by endocytosis. Invest Ophthalmol Vis Sci. 1999;40(11):2598-2607.
10. Carmichael A. Cytomegalovirus and the eye. Eye. 2012;26(2):237-240.
11. Hoover DR, Peng Y, Saah A, et al. Occurrence of cytomegalovirus retinitis after human immunodeficiency virus immunosuppression. Arch Ophthalmol. 1996;114(7):821-827.
12. Sugar EA, Jabs DA, Ahuja A, et al. Incidence of cytomegalovirus retinitis in the era of highly active antiretroviral therapy. Am J Ophthalmol. 2012;153(6):1016-1024.
13. Holland GN, Vaudaux JD, Shiramizu KM, et al. Characteristics of untreated AIDS-related cytomegalovirus retinitis. II. Findings in the era of highly active antiretroviral therapy (1997 to 2000). Am J Ophthalmol. 2008;145(1):12-22.
14. Jabs DA, Ahuja A, Van Natta M, et al. Course of cytomegalovirus retinitis in the era of highly active antiretroviral therapy: five-year outcomes. Ophthalmology. 2010;117(11): 2152-2161.
15. Kempen JH, Jabs DA, Wilson LA, et al. Risk of vision loss in patients with cytomegalovirus retinitis and the acquired immunodeficiency syndrome. Arch Ophthalmol. 2003;121(4):466-476.
16. Jabs DA, Ahuja A, Van Natta ML, et al. Long-term outcomes of cytomegalovirus retinitis in the era of modern antiretroviral therapy: results from a United States cohort. Ophthalmology. 2014;122(7):1452-1463.
17. Takakura A, Tessler HH, Goldstein DA, et al. Viral retinitis following intraocular or periocular corticosteroid administration: a case series and comprehenesive review of the literature. Ocul Immunol Inflamm. 2014;22(3):175-182.
18. Vadalpudi AD, Vadlapatla RK, Mitra AK. Current and emerging antivirals for the treatment of cytomegalovirus retinitis: an update on recent patents. Recent Pat Antiinfect Drug Discov. 2012;7(1):8-18.
19. Trivedi D, Williams RY, O’Reilly RJ, et al. Generation of CMV-specific T lymphocytes using protein-spanning pools of pp65-derived overlapping petadecapeptides for adoptive immunotherapy. Blood. 2005;105(7):2793-2801.
20. Koehne G, Hasan A, Doubrovina E, et al. Immunotherapy with donor T cells sensitized with overlapping pentadecapepetides for treatment of persistent cytomegalovirus infection or viremia. Biol Blood Marrow Transplant. 2014;21(9):1663-1678.
21. Gupta MP, Coombs P, Prockop SE, et al. Treatment of cytomegalovirus retinitis with cytomegalovirus-specific T-lymphocyte infusion. Ophthalmic Surg Lasers Imaging Retina. 2014;46(1):80-82.
Mrinali Gupta, MD
• assistant professor of ophthalmology at Weill Cornell Medical College in New York, N.Y.
• financial interest: none acknowledged
Szilárd Kiss, MD
• director of clinical research, director of compliance, and an associate professor of ophthalmology at Weill Cornell Medical College in New York, N.Y.; associate attending physician at the New York Presbyterian Hospital
• member of the Retina Today editorial advisory board
• financial disclosure: Weill Cornell Medical College and Dr. Kiss have intellectual property related to topic discussed in this article
• +1-646-962-2217; szk7001@med.cornell.edu











_1773249222.png?auto=compress,format&w=75)










