
Proliferative vitreoretinopathy (PVR) is a challenging complication of retinal detachment (RD) repair. PVR extending anteriorly can result in worse visual outcomes and hypotony.1 In this case, I present another complication induced by anterior PVR along with my approach for surgical repair.
Case Report
An 82-year-old man with hand-motion vision and intraocular pressure of 2 mm Hg was referred to me for management of a recurrent PVR-traction RD 4 weeks after a combined scleral buckle and vitrectomy for a macula-involving RD with pre-existing PVR. The patient was also found to have a dislocated plate-haptic IOL that coincided with his recurrent detachment.
Although I perform almost all surgeries with a 25-gauge approach, I elected for 23-gauge in this case. In my hands, plate-haptic IOLs are difficult to manipulate, and I wanted the additional grasping ability of the wider 23-gauge forceps platforms.
With a standard 3-port approach, I removed the 50% residual C3F8 gas bubble and placed a small bubble of perfluorocarbon liquid (PFCL) (Perfluoron, Alcon) to flatten the posterior pole and float the dislocated IOL to protect the macula. There were no significant posterior membranes, and I peeled the inferior equatorial preretinal membranes using ILM forceps (Figure 1). These membranes were well-defined and peeled easily.
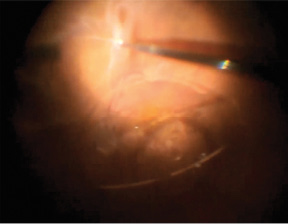
Figure 1. After a small bubble of PFCL is used to stabilize the posterior pole and float the IOL to protect the macula, forceps are used to peel inferior preretinal membranes.
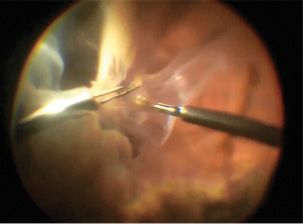
Figure 2. Massive pars plana membranes are peeled circumferentially using a bimanual approach.
As I continued peeling anteriorly, I found the membranes extended well into and circumferentially around the pars plana. The membranes were spongy and massive, and I placed a chandelier to enable a bimanual approach to peel them efficiently with a hand-over-hand technique (Figure 2).
My preference under these circumstances is to use MaxGrip forceps (Alcon), but they were not available on that day, and I used end-grasping forceps in one hand and ILM forceps in the other, which provided more than sufficient grip. As the peeled membranes accumulated, I used the vitreous cutter to remove them, and the larger gauge 23-gauge cutter was useful to easily digest these dense membranes.
In addition to a bimanual approach with two forceps, I employed a bimanual approach with forceps and the vitreous cutter, enabling me to efficiently digest and remove the membranes without further instrument exchange. I also employed a unimanual “spaghetti twirl” technique to wrap membranes around the vitreous cutter. The massive extent of these membranes in the pars plana was the likely etiology for the hypotony owing to traction on the ciliary body, and I hypothesize they resulted in IOL dislocation because of traction on the capsular bag.
After extensive and complete membrane peeling, I turned my attention to the dislocated IOL. I used the vitreous cutter to remove the residual capsular bag. Plate-haptic IOLs are difficult to manipulate, as they do not have distinct haptics that can be grasped easily and “hooked” into the anterior chamber.
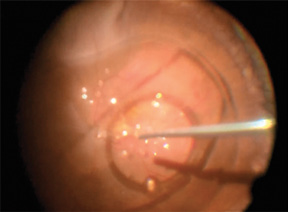
Figure 3. Additional PFCL is used to float the plate-haptic IOL anteriorly, allowing easy removal. Note the significant relaxation of the inferior retina following peeling.
After failed initial attempts to relocate the IOL into the anterior chamber, including using a bimanual approach with both forceps, I injected additional PFCL to provide internal drainage of the retinal detachment and to further float the IOL to near the iris plane (Figure 3). From there, it was easy to pop the IOL into the anterior chamber and remove it through a corneal incision. Of note, I had thought I would be able to cut the IOL and remove it through a small corneal incision, but I was not able to cut the rigid optic.
Plate-haptic IOLs are not commonly inserted, and I continue to struggle with their removal. What now works best for me is to engage one of the eyelets with the cutter or forceps to position it in the anterior vitreous followed by a hand-off to a second instrument passing through an already created scleral tunnel full-width incision to re-engage the same eyelet and remove it. I no longer try to position these IOLs first in the anterior chamber or cut them, and I use a 25-gauge approach.
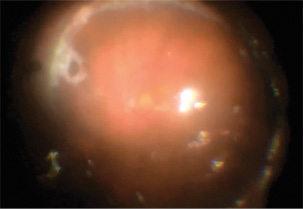
Figure 4. The retina is nicely attached after fluid-air exchange and focal laser. Silicone oil was subsequently placed.
Following closure of the corneal incision, the remainder of the case was uneventful. I performed a fluid-air exchange, treated the pre-existing tractional break with the laser, and filled the eye with silicone oil (Figure 4). The retina was relaxed and fully mobile following peeling, and although gas may have been sufficient, I thought oil was safest for anterior PVR.1 I placed an inferior iridectomy in this now aphakic eye.
Postoperatively, the patient developed extensive recurrent proliferation on the retinal surface, but fortunately the retina remained attached. I peeled these recurrent membranes and placed an ACIOL at the time of silicone oil removal 4 months later. The patient’s visual acuity stabilized at 20/200, and his eye remained normotensive without further recurrence of significant membranes.
Conclusion
We typically consider PVR in the setting of traction-inducing recurrent RD, whose repair is challenging. PVR membranes are not limited to the retina. They can extend into the pars plana and are commonly associated with hypotony, as in this case. These anterior membranes can also exert traction on the capsular bag, which may result in IOL dislocation in the setting of an already open capsule, as in this case.
Effectively addressing PVR membranes requires adequate visualization and creative approaches for complete peeling. Dyes to help stain PVR membranes and/or advanced visualization techniques such as scleral depression-assisted membrane peeling or endoscopic peeling may be required. Persistent challenges in visualization and complete peeling contribute to undue failure of these procedures.
IOL dislocation adds another set of challenges, particularly with plate-haptic IOLs that are difficult to manipulate. Floating the IOL with PFCL can be helpful. Without PFCL, I now take advantage of the eyelets in a “hand-off” technique to externalize the IOL. I look forward to learning others’ more elegant approaches.
The most important element of surgical success is appropriate planning. In this case, a 23-gauge approach, in contrast to my standard 25-gauge approach, was helpful to manipulate the plate-haptic IOL and to quickly digest the dense membranes and capsular bag. Poor planning and relative inexperience on my part added to difficulties with removing the plate-haptic IOL. I continue to learn from every case. n
Editorially independent supported by advertising from Alcon and Topcon.
1. Diddie KR, Azen SP, Freeman HM, et al. Anterior proliferative vitreoretinopathy in the silicone study. Silicone Study Report Number 10. Ophthalmology. 1996;103:1092-1099.
Paul Hahn, MD, PhD
• practices at NJRetina, Teaneck, NJ
• consultant for Second Sight Medical Products and Genentech, Inc.
• paulhahn@gmail.com





_1773249222.png?auto=compress,format&w=75)










