


The Boston Keratoprosthesis (KPro) is an effective surgical treatment for corneal blindness in patients who are not candidates for traditional penetrating keratoplasty, including those with severe ocular surface disease, such as Stevens-Johnson syndrome, ocular burns with limbal stem cell deficiency, or a history of multiple graft failures.
Worldwide, the Boston KPro is the most commonly used device of its kind.1 The type 1 KPro consists of a polymethylmethacrylate optic, which is placed through a ring of donor corneal tissue and held in place with a backing plate. This assembly is sutured to the host eye in a fashion identical to a standard penetrating keratoplasty. A plano device is available for implantation in pseudophakic eyes; and aphakic refractive powers are also available. A type 2 KPro, in which the optic penetrates the eyelid with a permanent tarsorrhaphy in place, is also available for cases of end-stage ocular surface disease.2,3
While the KPro can provide profound vision rehabilitation in eyes that might not otherwise be salvageable, it is commonly associated with vitreoretinal complications, including endophthalmitis or sterile vitritis, proliferative vitreoretinopathy, hypotony, retinal detachment, and choroidal detachment. The most common complication is the formation of retroprosthetic membranes, occurring in as many as 77% of cases. Additionally, many KPro eyes develop comorbid, medically refractive glaucoma resulting from the severity of their underlying disease.1
We present a case of a complex retinal detachment in a patient with a KPro who has concurrent glaucoma requiring silicone oil tamponade.
Case Report
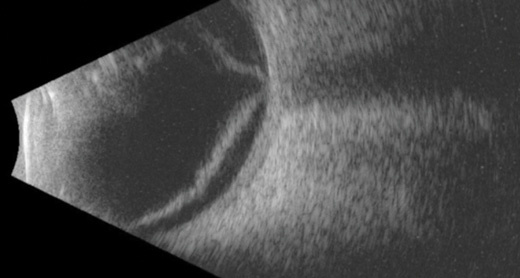
Figure 1. Horizontal B-scan ultrasound confirming macula-off retinal detachment.
An 82-year-old man with a history of Stephens-Johnson disease and an aphakic type 1 Boston KPro underwent pars plana vitrectomy and vitreous chamber insertion of a Baerveldt 250 glaucoma drainage implant (Abbott Laboratories, Inc.). As a result of inferior and nasal retinal detachment, his visual acuity had declined from 20/200 to finger counting. B-scan ultrasound confirmed that the macula was detached (Figure 1). A retroprosthetic membrane was also noted.
Procedure
Twenty-three-gauge vitrectomy was performed using the Constellation Vision System (Alcon) under monitored anesthesia care with retrobulbar block. An A.V.I. Panoramic Lens (Advanced Visual Instruments) was used for wide-field viewing through the KPro. Release of the posterior hyaloid was confirmed after core vitrectomy.
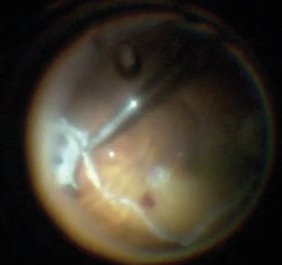
Figure 2. Creating a 180-degree inferior relaxing retinotomy using the vitreous cutter after demarcation with diathermy.
Perfluorocarbon liquid (PFCL) (Perfluoron, Alcon) was used to flatten and stabilize the posterior retina. The surgeon used bimanual technique with a 23-gauge lighted retinal pick (Alcon) and MaxGrip forceps (Alcon) to peel preretinal membranes completely off the retinal surface. All retinal breaks were identified. After maximal membrane peeling, intraretinal contraction was observed. Therefore, a 180-degree inferior relaxing retinotomy was outlined with diathermy then completed using the vitreous cutter (Figure 2). The anterior retinal tissue was removed, and the retina relaxed nicely under PFCL.
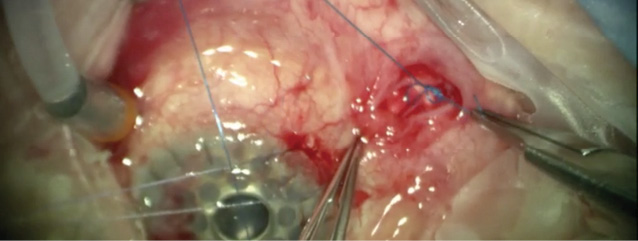
Figure 3. Ligation of the glaucoma drainage tube using Prolene suture.
Next, attention was turned to ligation of the Baerveldt shunt, as we anticipated silicone oil tamponade and did not want egress through the device, which would damage its mechanism. Westcott scissors and forceps were used to incise the superotemporal conjunctiva and expose the glaucoma drainage tube, which was then ligated using Prolene suture (Ethicon) to prevent efflux of silicone oil into the orbit (Figure 3).
A 25-gauge microvitreoretinal (MVR) blade was used to incise the retroprosthetic membrane, which was removed using MaxGrip forceps and the vitrectomy hand piece. Endolaser was applied with the retina stabilized under a complete fill of PFCL, after which a direct PFCL-to-silicone oil exchange was performed. One week postoperatively, the retina remained attached, and visual acuity had improved to 20/160.
Discussion
This case demonstrates that despite the presence of retroprosthetic membranes and small 3.3 mm diameter effective pupil size, good visualization of the posterior segment is possible with wide-field surgical viewing systems in patients with permanent Boston keratoprostheses. The retroprosthetic membrane may be removed with standard vitrectomy techniques, but the recurrence rate is high. Nd:Yag membranectomy can also be used to clear visually significant membranes, but this technique has been reported to increase the subsequent risk of retinal detachment four-fold.1
The advanced vitreoretinal pathology in KPro eyes is often best handled effectively and efficiently using bimanual techniques. Vitrectomy ports can be placed 1 mm to 3.5 mm posterior to the limbus, if it can be identified. Alternatively, making the sclerotomies at a radius 4.5 mm from the center of the optic places them just posterior to the KPro’s back plate. D’Amico and Ray et al., report successful vitrectomies were performed using incisions made directly through the eyelid, conjunctiva, and sclera using an MVR blade in patients with type 2 Boston KPros.4,5
Long-term silicone oil tamponade may be necessary to stabilize the retina during healing and to prevent hypotony and phthisis.1 In this case, the presence of a glaucoma drainage implant in the vitreous cavity could have led to problems from silicone oil tamponade, as migration of silicone oil into such devices, the subconjunctival space, and the orbit has been reported.6,7 This can result in granulomatous inflammation, pain, orbital mass effects, and impaired function of adjacent structures.8 These complications can be avoided by ligating or removing the drainage tube.
As demonstrated in this case, direct PFCL-to-silicone oil exchange stabilizes the retinectomy edge without risk of slippage compared to a fluid-air then air-silicone oil exchange.
Conclusion
The Boston KPro is an effective treatment for corneal blindness, but it is often used in eyes with comorbid glaucoma and can be associated with vitreoretinal complications. While these eyes pose a challenge to the vitreoretinal surgeon, wide-field visualization through the device is possible and advanced bimanual techniques can be employed effectively in the management of posterior segment complications. n
Editorially independent supported by advertising from Alcon and Topcon.
1. Klufas MA, Yannuzzi NA, D’Amico DJ, Kiss S. Vitreoretinal aspects of permanent keratoprosthesis. Surv Ophthalmol. 2015;60:216-228.
2. Doane MG, Dohlman CH, Bearse G. Fabrication of a keratoprosthesis. Cornea.1996;15:179-184.
3. Dohlman CH, Harissi-Dagher M, Graney J. The Boston Keratoprosthesis: a new threadless design. Digital J Ophthalmol. 2007;13(3).
4. D’Amico DJ. The Gertrude D. Pyron 2016 Award Lecture of the American Society of Retina Specialists: Permanent Keratoprosthesis and Complex Ocular Reconstruction: New Opportunities and Challenges for the Vitreoretinal Surgeon. J VitreoRetinal Diseases. May 2017;1(3).
5. Ray S, Khan BF, Dohlman CH, D’Amico DJ. Management of vitreoretinal complications in eyes with permanent keratoprosthesis. Arch Ophthalmol. 2002;120:559-566.
6. Nazemi PP, Chong LP, Varma R, Burnstine MA. Migration of intraocular silicone oil into the subconjunctival space and orbit through an Ahmed glaucoma valve. Am J Ophthalmol. 2001;132:929-931.
7. Friberg TR, Fanous MM. Migration of intravitreal silicone oil through a Baerveldt tube into the subconjunctival space. Semin Ophthalmol. 2004;19:107-108.
8. Couch SM, Harocopos GJ, Holds JB. Orbital silicone oil granuloma discovered during enucleation. Arch Ophthalmol. 2012 Aug;130:1083-1085.
Zachary M. Bodnar, MD
• first year vitreoretinal surgery fellow at the Byers Eye Institute at Stanford
• no relevant financial disclosures
• zbodnar@stanford.edu
Francisco A. Folgar, MD
• practices at Carolina Eyecare Physicians, Mount Pleasant, SC
• no relevant financial disclosures
Prithvi Mruthyunjaya, MD, MHS
• Associate Professor of ophthalmology at Stanford University, Stanford, CA
• Director of Ocular Oncology at the Byers Eye Institute, Palo Alto, CA
• consultant: Optos, SPARK therapeutics, Santen, Castle Biosciences
• prithvi9@stanford.edu





_1773249222.png?auto=compress,format&w=75)










