OCT angiography (OCTA) is a new, rapid, noninvasive imaging modality. It uses motion contrast to visualize retinal and choroidal vasculature without the use of extrinsic dyes. OCTA images are obtained by decorrelating successive OCT B-scans acquired in the same area. Because the only change between successive OCT scans of the same retinal location should be due to the movement of blood through the vessels, OCTA software generates a map of the vasculature in the back of the eye.1,2
As a derivative of OCT, this new modality also delivers high-resolution, depth-resolved images (Figure 1), which is an advantage over fluorescein angiography (FA) and indocyanine green angiography (ICGA). The high resolution allows visualization of the microvasculature in much greater detail compared with dye-based angiography, potentially in a quantitative manner. Because of the depth resolution, individual vascular layers can be visualized independently.1 These characteristics have quickly led OCTA to become vital in the evaluation of patients with several chorioretinal pathologies.

Figure 1. OCTA en face projections of full retinal depth (A), superficial retinal slab (B), and deep retinal slab (C), acquired in a healthy individual. Segmented B-scans with flow overlay corresponding with each en face image are shown on the right (D-F).
Two forms of OCTA are in use: spectral-domain (SD) and swept-source (SS) OCTA. An advantage of SS-OCTA over SD-OCTA is its faster scanning speed, which makes possible larger scan areas and denser scan patterns with comparable acquisition times. Additionally, SS-OCTA has less sensitivity roll-off with depth, and it typically uses a longer wavelength; these characteristics allow better visualization through opacities and visualization of deeper structures.3,4
In this report, we outline the clinical utility of OCTA in exudative and nonexudative AMD. In Part Two of this two-part series we will examine its usefulness for other retinal and choroidal vascular diseases.
OCTA IN AMD
AMD is the leading cause of vision loss in developed countries.5 Severe vision loss in advanced AMD is associated with two conditions: geographic atrophy (GA) and macular neovascularization (MNV). OCTA has clinical utility in both the dry and wet forms of AMD.6
Dry AMD
In patients with dry AMD, the primary utility of OCTA is in identifying eyes that are phenotypically dry but that have underlying nonexudative neovascular disease. Nonexudative MNV has been described as type 1 neovascularization without exudative retinal changes. These lesions can be seen as staining plaques on ICGA. They display no leakage on FA or subretinal fluid on OCT.7
OCTA assists in the diagnosis of nonexudative MNV by identifying the presence of flow underneath the retinal pigment epithelium (RPE) and above Bruch membrane, in a flat irregular pigment epithelial detachment (PED; Figure 2).7
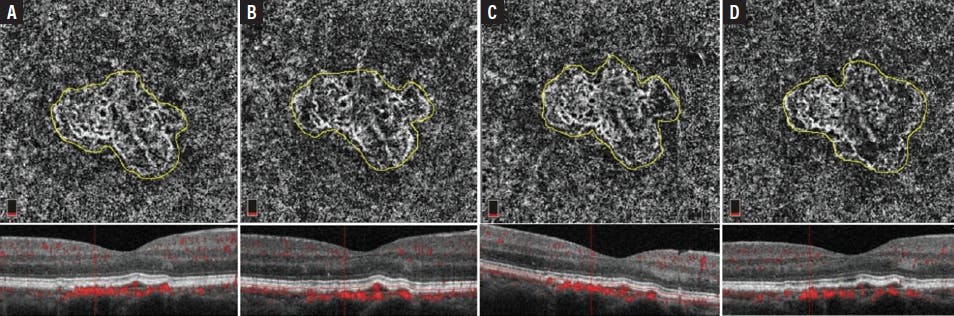
Figure 2. OCTA en face images (top row) and B-scans (bottom row) from an eye with nonexudative MNV: follow-up through 4 consecutive years (A-D). The MNV lesion area has increased in size from 1.445 mm2 at baseline (A) to 1.620 mm2 on the most recent visit (D).
Roisman et al used a multimodal approach to follow 11 patients with intermediate nonexudative AMD in one eye and type 1 neovascular AMD in the fellow eye. Three of 11 patients showed a plaque lesion on ICGA correlating with subclinical MNV detected by OCTA.8 Another study found that eyes with subclinical MNV have a higher risk of exudation after 1 year of follow-up compared with eyes without detectable subclinical MNV at baseline. Although there is no evidence of benefit in treating these patients, they represent a higher-risk cohort that perhaps warrants closer follow-up.9
Wet AMD
One of the most important applications of OCTA is in the detection of neovascularization in wet AMD.4 En face OCTA images can be used to visualize vascularized networks, while their exact location in the retina is depicted by B-scans.4,7
MNV on OCTA has been described as a central trunk vessel with a peripheral capillary tuft, although this morphology is not applicable to all MNVs. Several other morphology terms have been proposed, many lacking validation.10 Compared with FA, MNV lesions captured on OCTA tend to be smaller, probably due to better delineation of the neovascular complex margins on OCTA, which can be obscured by leakage on FA.11
The sensitivity and specificity of OCTA compared with FA and ICGA vary across studies, with a range of 50% to 100%.12 It should be noted that published studies have used different devices, and some authors have included a mix of MNV subtypes.12
Shadowing due to hemorrhage and the presence of tall PEDs can increase the false negative rate. Combined application of OCTA and cross-sectional OCT has helped to improve sensitivity and specificity in detecting type 1 MNVs. The detection of type 3 MNV and polypoidal complexes seems to be more challenging on OCTA.12
Although OCTA can be used to monitor MNV lesion response to anti-VEGF treatment, studies on this topic have not been in full agreement. Whereas some authors reported a reduction in MNV size,13 others found no change or even an increase in size after 1 to 2 weeks of maximal regression (Figure 3). It has also been demonstrated that, after anti-VEGF treatment, MNV may lose some of the fibrillary vessels at the edges.1,4,14

Figure 3. Morphologic aspect of MNV before and after treatment. The OCTA en face image (A) and B-scan (B) show the MNV lesion before injection. The OCTA en face image (C) and B-scan (D) show the MNV lesion 5 weeks after injection.
Care should be taken regarding common OCTA interpretation pitfalls that can arise from either image acquisition or processing. One such error occurs in the presence of GA and RPE disruption, causing enhanced signal penetration into the choroid. With concurrent loss of choriocapillaris, large choroidal vessels can be displaced upward and mimic an MNV on en face OCTA. B-scan with and without flow overlay will show increased choroidal flow and signal hypertransmission, respectively, the latter of which is a notable characteristic of GA.4
Retinal pathologies can disrupt retinal layer detection, automated segmentation, and, consequently, the preset slabs. On these occasions, retinal slab segmentation should be performed manually to ensure correct detection of retinal boundaries.4
CONCLUSION
OCTA is a rapid, noninvasive imaging tool useful for a wide range of ophthalmic diseases in the clinical setting. However, limitations such as artifacts and segmentation errors can make scan interpretation challenging.15 The inability to detect leakage is also a deficit compared with FA, limiting the clinical utility of OCTA. However, the higher resolution and the depth-resolved property of OCTA can add valuable information to clinical assessments of many conditions.1
1. Spaide RF, Fujimoto JG, Waheed NK, Sadda SR, Staurenghi G. Optical coherence tomography angiography. Prog Retin Eye Res. 2018;64:1-55.
2. de Carlo TE, Romano A, Waheed NK, Duker JS. A review of optical coherence tomography angiography (OCTA). Int J Retina Vitreous. 2015;1:5.
3. Miller AR, Roisman L, Zhang Q, et al. Comparison Between Spectral-Domain and Swept-Source Optical Coherence Tomography Angiographic Imaging of Choroidal Neovascularization. Invest Ophthalmol Vis Sci. 2017;58(3):1499-1505.
4. Greig EC, Duker JS, Waheed NK. A practical guide to optical coherence tomography angiography interpretation. Int J Retina Vitreous. 2020;6(1):55.
5. Wong WL, Su X, Li X, et al. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. Lancet Glob Health. 2014;2(2):e106-e116.
6. Chalam KV, Sambhav K. Optical Coherence Tomography Angiography in Retinal Diseases. J Ophthalmic Vis Res. 2016;11(1):84-92.
7. Querques G, Srour M, Massamba N, et al. Functional characterization and multimodal imaging of treatment-naive “quiescent” choroidal neovascularization. Invest Ophthalmol Vis Sci. 2013;54(10):6886-6892.
8. Roisman L, Zhang Q, Wang RK, et al. Optical Coherence Tomography Angiography of Asymptomatic Neovascularization in Intermediate Age-Related Macular Degeneration. Ophthalmology. 2016;123(6):1309-1319.
9. de Oliveira Dias JR, Zhang Q, Garcia JMB, et al. Natural History of Subclinical Neovascularization in Nonexudative Age-Related Macular Degeneration Using Swept-Source OCT Angiography. Ophthalmology. 2018;125(2):255-266.
10. Mendonça LSM, Perrott-Reynolds R, Schwartz R, et al. Deliberations of an International Panel of Experts on OCTA Nomenclature of nAMD [published online ahead of print, 2020 Dec 25]. Ophthalmology.
11. Costanzo E, Miere A, Querques G, Capuano V, Jung C, Souied EH. Type 1 Choroidal Neovascularization Lesion Size: Indocyanine Green Angiography Versus Optical Coherence Tomography Angiography. Invest Ophthalmol Vis Sci. 2016;57(9):OCT307-OCT313.
12. Perrott-Reynolds R, Cann R, Cronbach N, et al. The diagnostic accuracy of OCT angiography in naive and treated neovascular age-related macular degeneration: a review. Eye (Lond). 2019;33(2):274-282.
13. Kuehlewein L, Sadda SR, Sarraf D. OCT angiography and sequential quantitative analysis of type 2 neovascularization after ranibizumab therapy. Eye (Lond). 2015;29(7):932-935.
14. Amoroso F, Miere A, Semoun O, Jung C, Capuano V, Souied EH. Optical coherence tomography angiography reproducibility of lesion size measurements in neovascular age-related macular degeneration (AMD). Br J Ophthalmol. 2018;102(6):821-826.
15. Arya M, Sabrosa AS, Duker JS, Waheed NK. Choriocapillaris changes in dry age-related macular degeneration and geographic atrophy: a review. Eye Vis (Lond). 2018;5:22. Published 2018 Sep 15.













_1773249222.png?auto=compress,format&w=75)












