AT A GLANCE
- New imaging tools such as flavoprotein fluorescence technology, laser Doppler holography, photoacoustic imaging, and transscleral optical phase imaging are changing the way researchers see the retina and document ocular diseases.
- Adaptive optics OCT has vastly improved and can be used for in vivo assessment of the retinal vasculature, cone function and density, and retinal pigment epithelium cell density.
- Super-resolution MRI, which can produce high-resolution images of the eye and lower scan times significantly, is particularly useful for myopic eyes.
Advanced ocular imaging has transformed clinical eye care and is now integrated into the early detection, accurate diagnosis, and appropriate management of a variety of ocular diseases. As new imaging modalities emerge that tout advances in analytics, optics, and lasers, researchers are investigating their clinical utility before they become a part of routine practice. Here are some of the tools working their way through the ophthalmic imaging pipeline.
FLUORESCENCE IMAGING
The OcuMet Beacon (OcuSciences) identifies retinal metabolic diseases using flavoprotein fluorescence (FPF) technology. The device shines blue light onto the retina to excite flavoproteins, which show a green autofluorescence.1 The amount of fluorescence emitted by the flavoprotein indicates reversible metabolic stress due to disease, before cell death. The more intense the signal output the device generates, the more metabolic dysfunction it is measuring.
Conversely, a decrease in metabolic stress may reduce FPF intensity. In a study by Romo et al, eight patients with diabetic retinopathy (DR), diabetic macular edema (DME), and/or clinically significant DME (CSDME) were imaged before and after treatment with anti-VEGF therapy.2 The results showed a strong correlation between decreased FPF and improvement in BCVA following therapy (Figure 1). One patient with DME (not CSDME) had significantly improved FPF after anti-VEGF treatment, demonstrating that this imaging modality may detect mechanisms not due to CSDME.2
![<p>Figure 1. The fundus images of a 65-year-old Hispanic male diagnosed with proliferative DR and CSDME before (A) and after (B) one injection of an anti-VEGF agent show macular edema, hard exudates, and hemorrhages. OCT imaging shows a central macular thickness of 574 μm before treatment (C) and 541 μm after injection (D). The FPF map shows an increase in the green-yellow coloring before anti-VEGF therapy (E), suggesting greater mitochondrial dysfunction (oxidative stress) compared with the blue-green FPF map after injection (F). [gsu = grayscale units] Reprinted with permission from Hindawi Pub. Corp.<sup>2</sup></p>](https://core4-cms.imgix.net/issue-2207/1122_F5_Fig1.png?auto=compress,format)
Figure 1. The fundus images of a 65-year-old Hispanic male diagnosed with proliferative DR and CSDME before (A) and after (B) one injection of an anti-VEGF agent show macular edema, hard exudates, and hemorrhages. OCT imaging shows a central macular thickness of 574 μm before treatment (C) and 541 μm after injection (D). The FPF map shows an increase in the green-yellow coloring before anti-VEGF therapy (E), suggesting greater mitochondrial dysfunction (oxidative stress) compared with the blue-green FPF map after injection (F). [gsu = grayscale units] Reprinted with permission from Hindawi Pub. Corp.2
The OcuMet Beacon can detect the presence of FPF at the optic nerve head, indicating mitochondrial dysfunction and glaucomatous damage.3 Ahsanuddin et al found that FPF values were inversely correlated with BCVA and elevated in conditions known to be mediated by oxidative stress, such as retinal vein occlusion, AMD, and proliferative DR.4
Fluorescence lifetime imaging ophthalmoscopy (FLIO) monitors changes in the retina using a similar method—measuring the lifetimes of fundus autofluorescence. Using FLIO, clinicians can measure the time a molecule spends in an excited state before returning to a ground state. In addition to standard intensity images, FLIO creates detailed characterizations of ocular diseases, such as choroideremia, Stargardt disease, and macular telangiectasia type 2, through hyperfluorescent lesions or hypofluorescent atrophic areas.5-7
LASER DOPPLER HOLOGRAPHY
Laser Doppler holography (LDH) is a digital holographic imaging modality that is capable of measuring retinal and choroidal blood flow with ultrafast frame rates (a few milliseconds) and high temporal resolution.8,9 LDH analyzes the frequency spectrum between the monochromatic reference beam and the Doppler broadened beam to create blood flow contrast.10,11 The holographic technique allows for higher speed acquisition fields and can work with very low exposure time.12,13
In a study by Puyo et al, the high temporal resolution of LDH showed the pulsatile nature of blood flow through the retina.8 The data showed a full-field temporal resolution of 1.6 ms while maintaining the retinal exposure under 1.5 mW at 785 nm. A prior study found that LDH displayed structures in the choroidal vasculature not previously observed with OCT and ICG angiography instruments.14 LDH can also identify retinal and choroidal arteries and veins unambiguously based on the variation in systole and diastole and pulsatile flow measurements.15-17
In another study by Puyo et al, LDH effectively imaged blood flow in the bulbar conjunctiva and episclera and documented anterior segment transparency over the entrance pupil with a single image, demonstrating simultaneous imaging of the anterior and posterior segments. Compared with slit lamp examinations, the authors found that LDH provided better contrast of opacities, is highly repeatable and comfortable for patients, and is not operator-dependent.18
VISIBLE LIGHT OCT
Visible light OCT (vis-OCT) uses a supercontinuum light source—rather than near-infrared (NIR) light—to improve the imaging resolution and retrieve unique tissue scattering and absorption.19 Vis-OCT has a higher axial resolution than NIR-based OCT with comparable bandwidth; it also provides an increased imaging contrast due to higher scattering coefficients for spectroscopic analysis (Figure 2).19-21
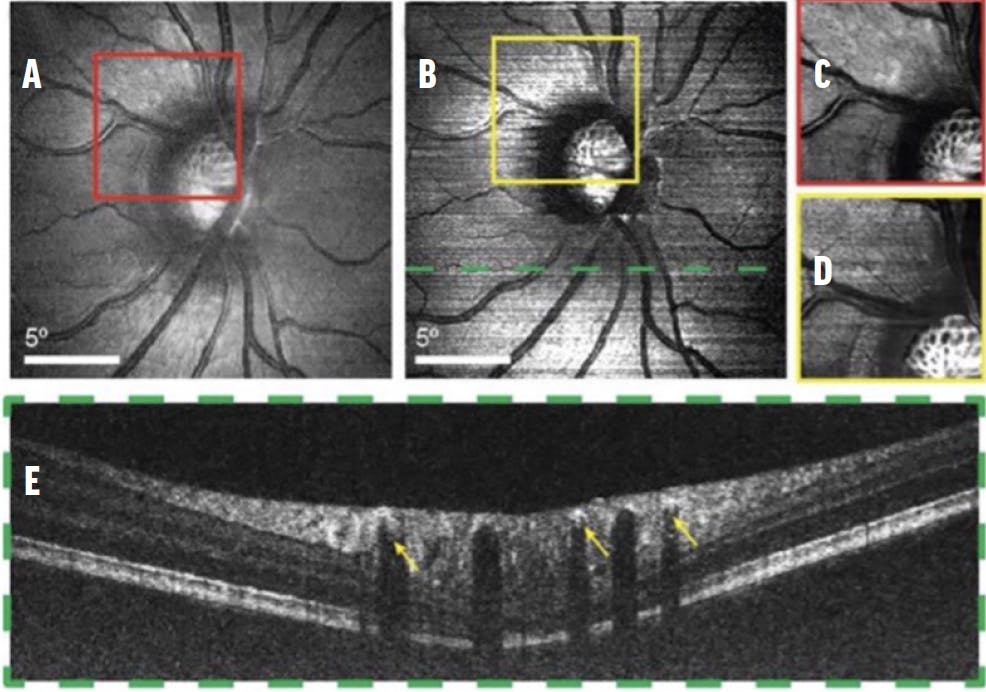
Figure 2. These SLO (A) and vis-OCT images (B)—contrast-adjusted in C and D, respectively—are centered at the optic nerve head of the same patient. In the cross-sectional vis-OCT image (E) from the location highlighted in B, the arrows point to the signal from the blood within major retinal vessels. Reprinted with permission from Optica.22
Vis-OCT provides imaging of all four layers of the outer bands of the retina. Zhang et al used vis-OCT to investigate contributors to the outermost (4th) hyperreflective band, and they found that melanosomes are the predominant contributors to retinal pigment epithelium (RPE) backscattering in young healthy eyes.22
The device has been shown to be suitable for routine clinic visits, and researchers have used a portable vis-OCT system to provide high-quality images of the retina in diseased and healthy eyes.23 More recently, vis-OCT has been combined with NIR to validate the system’s ability to detect structural abnormalities in en face and B-scan images.24 Vis-OCT can also measure retinal oxygen metabolic rate and facilitate oximetry by using a narrower bandwidth spectrometer, creating vis-OCT angiography.21,25
ADAPTIVE OPTICS OCT
Adaptive optics OCT (AO-OCT) is a research tool with only a few systems available worldwide. The technology has been used for in vivo assessment of the retinal vasculature, cone function and density, and RPE cell density.26 Imaging using AO-OCT is primarily performed in the macular area where light backscattered from the photoreceptor band creates a signal, resulting in a reliable wavefront measurement.
Until recently, AO-OCT imaging was mostly used in mice. Pollreisz et al reported the first successful use of AO-OCT with a fundus camera to examine patients with early and late AMD in a routine clinical setting.27 In addition, the device has 3D capabilities and can visualize single photoreceptors, the lamina cribosa, and retinal nerve fiber bundles.28-30
PHOTOACOUSTIC IMAGING
Photoacoustic imaging (PAI) uses the absorption characteristics of endogenous or exogenous biomarkers to generate chromophore selective image contrast.31,32 This noncontact technology uses both light and sound waves to detect activity at the molecular level and is typically paired with OCT due to its detailed scatter information. PAI provides metabolic and molecular information, and in combination with spectral-domain OCT, has been used to evaluate microvasculature flow states. The complementary nature of PAI and OCT may be useful in studying diseases related to the functional blood supply.33
Multi-wavelength PAI microscopy has been combined with swept-source OCT to demonstrate both systems’ ability to create imaging contrasts of optical scatter and absorption.34 A study by Hosseinaee et al showed that PAI/OCT can provide noninvasive, accurate measurements of functional details in ophthalmic tissue (Figure 3).34 Other researchers have used PAI/OCT to image choroidal neovascularization and RPE cells in the setting of regenerative cell therapies.35,36
More research is needed, as the PAI scanning speed does not yet reach that of the OCT system in all scenarios.33 Some studies have addressed this using the stimulated Raman scattering effect to implement high-speed, multi-wavelength light sources.34,37

Figure 3. In vivo imaging of the ocular tissue using a multimodal photoacoustic remote sensing (PARS) OCT system shows a volume OCT rendering of the eye (A). The OCT B-scan represents a cross-section of the iris vasculature (B, yellow arrows). The en face OCT image (C) and vasculature of the iris are imaged with the PARS subsystem over the dashed area (D) with overlayed PARS and OCT images (E). Reprinted with permission from Nature Publishing Group; Springer Science and Business Media. 35
TRANSSCLERAL OPTICAL PHASE IMAGING
Combined with adaptive optics, transscleral optical phase imaging (TOPI) uses high-angle oblique illumination of the retina to enhance cell contrast. Through TOPI, clinicians can visualize RPE and nerve fiber layer cells over a large field of view, without the downside of long exposure time. An oblique beam of light crosses the sclera and scatters to create a divergent beam that illuminates the posterior retina evenly (Figure 4).38,39

Figure 4. The yellow box on the SLO imaging of a volunteer’s left eye (A) illustrates the position of the TOPI image of the RPE layer (B). The individual RPE cells appear as dark spots with bright edges. Reprinted with permission from Nature Research; Springer Science and Business Media.40
TOPI can provide a noninvasive quantitative assessment of the RPE and choriocapillaris with fewer eye-motion artifacts due to the fast acquisition time.40,41 Unlike adaptive optics scanning laser ophthalmoscopy (SLO) and AO-OCT, which may be more difficult to use in a clinical setting to achieve cellular-resolution imaging, TOPI uses a similar mechanism but with high-angle oblique illumination of the retina.38 Kowalczuk et al found the technology to be safe and repeatable, as well as consistent with previous in vivo studies of RPE cells.42
SUPER-RESOLUTION MRI
Super-resolution MRI (srMRI), a novel tool designed to provide precise medical diagnosis and analysis, categorizes image patches as either smooth or textured. The srMRI denoises the patches using an algorithm that takes an estimated high-resolution image gradient profile to determine gradient profile sharpness and improve the signal-to-noise ratio.43
Hoang et al used srMRI to produce high-resolution (250-μm isotropic), 3D images of highly myopic eyes.44 The technology produced high-resolution images of the entire eye while also lowering scan times significantly and reducing motion artifacts.44 Another study of a different srMRI system also obtained high-resolution and contrast MRI images of the entire eye while avoiding significant motion artifacts.45 This device is particularly beneficial for myopic eyes because the retinal curvature imaged with 2D OCT imaging can be artificially flattened and misrepresent the true anatomy.46
NEXT STEPS
Over the past three decades, the emergence of new technology has revolutionized imaging modalities that document ocular health and disease. Knowledge gained from these innovative approaches can help guide the future of ocular disease screening, diagnosis, and management.
1. Ocumet. Ocumet Beacon. Accessed October 3, 2022. www.ocumet.com.
2. Andrade Romo JS, Lynch G, Liu K, et al. Flavoprotein fluorescence correlation with visual acuity response in patients receiving anti-VEGF injection for diabetic macular edema. Oxid Med Cell Longev. 2018;2018:3567306.
3. Zhou DB, Castanos MV, Geyman L, et al. Mitochondrial dysfunction in primary open-angle glaucoma characterized by flavoprotein fluorescence at the optic nerve head. Ophthalmol Glaucoma. 2022;5(4):413-420.
4. Ahsanuddin S, Otero-Marquez O, Rich C, Rosen RB. Flavoprotein fluorescence (FPF) elevation using retinal metabolic analysis correlates with decreased visual acuity in patients with various retinal pathologies. Invest Ophthalmol Vis Sci. 2022;63(7):3515-3515.
5. Vitale AS, Sauer L, Modersitzki NK, Bernstein PS. Fluorescence lifetime imaging ophthalmoscopy (FLIO) in patients with choroideremia. Transl Vis Sci Technol. 2020;9(10):33.
6. Dysli C, Wolf S, Hatz K, Zinkernagel MS. Fluorescence lifetime imaging in stargardt disease: potential marker for disease progression. Invest Ophthalmol Vis Sci. 2016;57(3):832-841.
7. Sauer L, Gensure RH, Hammer M, Bernstein PS. Fluorescence lifetime imaging ophthalmoscopy: a novel way to assess macular telangiectasia type 2. Ophthalmol Retina. 2018;2(6):587-598.
8. Puyo L, Paques M, Atlan M. Spatio-temporal filtering in laser Doppler holography for retinal blood flow imaging. Biomed Opt Express. 2020;11(6):3274-3287.
9. Puyo L, Paques M, Fink M, Sahel JA, Atlan M. Holographic laser Doppler retinal imaging. Invest Ophthalmol Vis Sci. 2018;59(9):5861-5861.
10. Magnain C, Castel A, Boucneau T, et al. Holographic laser Doppler imaging of microvascular blood flow. J Opt Soc Am A Opt Image Sci Vis. 2014;31(12):2723-2735.
11. Donnarumma D, Brodoline A, Alexandre D, Gross M. 4D holographic microscopy of zebrafish larvae microcirculation. Opt Express. 2016;24(23):26887-26900.
12. Hillmann D, Spahr H, Hain C, et al. Aberration-free volumetric high-speed imaging of in vivo retina. Sci Rep. 2016;6:35209.
13. Ginner L, Schmoll T, Kumar A, et al. Holographic line field en-face OCT with digital adaptive optics in the retina in vivo. Biomed Opt Express. 2018;9(2):472-485.
14. Puyo L, Paques M, Fink M, Sahel JA, Atlan M. Choroidal vasculature imaging with laser Doppler holography. Biomed Opt Express. 2019;10(2):995-1012.
15. Puyo L, Paques M, Fink M, Sahel JA, Atlan M. Waveform analysis of human retinal and choroidal blood flow with laser Doppler holography. Biomed Opt Express. 2019;10(10):4942-4963.
16. Simonutti M, Paques M, Sahel JA, et al. Holographic laser Doppler ophthalmoscopy. Opt Lett. 2010;35(12):1941-1943.
17. Puyo L, Paques M, Fink M, Sahel JA, Atlan M. In vivo laser Doppler holography of the human retina. Biomed Opt Express. 2018;9(9):4113-4129.
18. Puyo L, David C, Saad R, et al. Laser Doppler holography of the anterior segment for blood flow imaging, eye tracking, and transparency assessment. Biomed Opt Express. 2021;12(7):4478-4495.
19. Shu X, Beckmann L, Zhang H. Visible-light optical coherence tomography: a review. J Biomed Opt. 2017;22(12):1-14.
20. Song W, Zhang S, Kim YM, et al. Visible light optical coherence tomography of peripapillary retinal nerve fiber layer reflectivity in glaucoma. Transl Vis Sci Technol. 2022;11(9):28.
21. Yi J, Liu W, Chen S, et al. Visible light optical coherence tomography measures retinal oxygen metabolic response to systemic oxygenation. Light Sci Appl. 2015;4(9):e334.
22. Zhang T, Kho AM, Yiu G, Srinivasan VJ. Visible light optical coherence tomography (OCT) quantifies subcellular contributions to outer retinal band 4. Transl Vis Sci Technol. 2021;10(3):30.
23. Shu X, Beckmann L, Wang Y, et al. Designing visible-light optical coherence tomography towards clinics. Quant Imaging Med Surg. 2019;9(5):769-781.
24. Wang J, Baker A, Subramanian ML, et al. Simultaneous visible light optical coherence tomography and near infrared OCT angiography in retinal pathologies: A case study. Exp Biol Med (Maywood). 2022;247(5):377-384.
25. Song W, Shao W, Yi W, et al. Visible light optical coherence tomography angiography (vis-OCTA) facilitates local microvascular oximetry in the human retina. Biomed Opt Express. 2020;11(7):4037-4051.
26. Pircher M, Zawadzki RJ. Review of adaptive optics OCT (AO-OCT): principles and applications for retinal imaging. Biomed Opt Express. 2017;8(5):2536-2562.
27. Pollreisz A, Salas M, Sacu S, et al. Adaptive-optics (AO) optical coherence tomography (OCT) imaging of the photoreceptor (PR) layer in early and advanced dry age-related macular degeneration (AMD) in clinical routine. Invest Ophthalmol Vis Sci. 2017;58(8):1577.
28. Kocaoglu OP, Turner TL, Liu Z, Miller DT. Adaptive optics optical coherence tomography at 1 MHz. Biomed Opt Express. 2014;5(12):4186-4200.
29. Nadler Z, Wang B, Schuman JS, et al. In vivo three-dimensional characterization of the healthy human lamina cribrosa with adaptive optics spectral-domain optical coherence tomography. Invest Ophthalmol Vis Sci. 2014;55(10):6459-6466.
30. Kocaoglu OP, Cense B, Jonnal RS, et al. Imaging retinal nerve fiber bundles using optical coherence tomography with adaptive optics. Vision Res. 2011;51(16):1835-1844.
31. Yao J, Wang LV. Photoacoustic microscopy. Laser Photon Rev. 2013;7(5).
32. Wang LV, Hu S. Photoacoustic tomography: in vivo imaging from organelles to organs. Science. 2012;335(6075):1458-1462.
33. Ma Z, Luo S, Yu M, et al. Assessment of microvasculature flow state with a high speed all-optic dual-modal system of optical coherence tomography and photoacoustic imaging. Biomed Opt Express. 2018;9(12):6103-6115.
34. Hosseinaee Z, Nima A, Pellegrino N, Khalili L, Mukhangaliyeva L, Haji Reza P. Functional and structural ophthalmic imaging using noncontact multimodal photoacoustic remote sensing microscopy and optical coherence tomography. Sci Rep. 2021;11(1):11466.
35. Nguyen VP, Qian W, Li Y, et al. Chain-like gold nanoparticle clusters for multimodal photoacoustic microscopy and optical coherence tomography enhanced molecular imaging. Nat Commun. 2021;12(1):34.
36. Nguyen VP, Li Y, Henry J, et al. In vivo subretinal ARPE-19 cell tracking using indocyanine green contrast-enhanced multimodality photoacoustic microscopy, optical coherence tomography, and fluorescence imaging for regenerative medicine. Transl Vis Sci Technol. 2021;10(10):10.
37. Hajireza P, Forbrich A, Zemp R. In-vivo functional optical-resolution photoacoustic microscopy with stimulated Raman scattering fiber-laser source. Biomed Opt Express. 2014;5(2):539-546.
38. Laforest T, Künzi M, Kowalczuk L, Carpentras D, Behar-Cohen F, Moser C. Transscleral optical phase imaging of the human retina. Nat Photonics. 2020;14(7):439-445.
39. Künzi M, Laforest T, Kowalczuk L, Behar-Cohen FF, Moser C. Transscleral optical phase imaging (TOPI) of human retinal pigment epithelium. Invest Ophthalmol Vis Sci. 2019;60(9):4741.
40. Mantel I, Kowalcuk LE, Iskandar A, et al. Transscleral optical phase imaging revealing the choriocapillaris structure. Invest Ophthalmol Vis Sci. 2021;62(8):1911.
41. Caetano Dos Santos FL, Laforest T, Künzi M, Kowalczuk L, Behar-Cohen F, Moser C. Fully automated detection, segmentation, and analysis of in vivo RPE single cells. Eye (Lond). 2021;35(5):1473-1481.
42. Kowalczuk L, Dornier R, Kunzi M, et al. In vivo retinal pigment epithelium imaging using transscleral optical imaging in healthy eyes [published online ahead of print October 18, 2022]. Ophthalmol Retina.
43. Kaur P, Sao AK, Ahuja CK. Super resolution of magnetic resonance images. J Imaging. 2021;7(6):101.
44. Hoang QV, Chang S, Najjar R, et al. Novel super resolution magnetic resonance imaging processing pipeline for whole eye imaging with short scan times in high myopic eyes. Invest Ophthalmol Vis Sci. 2022;63(7):4334–A0039.
45. Glarin RK, Nguyen BN, Cleary JO, et al. MR-EYE: high-resolution MRI of the human eye and orbit at ultrahigh field (7T). Magn Reson Imaging Clin N Am. 2021;29(1):103-116.
46. Kuo AN, Verkicharla PK, McNabb RP, et al. Posterior eye shape measurement with retinal OCT compared to MRI. Invest Ophthalmol Vis Sci. 2016;57(9):OCT196-203.



















_1773249222.png?auto=compress,format&w=75)












