Choroidal folds are clinical entities in which subretinal striae are seen in the posterior pole or periphery of the retina. Choroidal folds rarely extend beyond the equator,1 and they present with undulations of the inner choroid, Bruch membrane, overlying retinal pigment epithelium (RPE), and neurosensory retina.2,3
This clinical finding is usually detected during routine ophthalmic examination of patients who are asymptomatic. It has been suggested that choroidal folds form when the Bruch membrane is forced to fold with choroidal expansion or swelling.1,2 Strain between the sclera and the choroid has also been proposed as a mechanism of choroidal fold development.4
Regardless of the cause, long-lasting choroidal folds can lead to RPE atrophy, Bruch membrane breaks/damage, and, eventually, choroidal neovascularization (CNV). They are usually horizontal but can also present in an oblique, vertical, circumferential, or irregular pattern.1,3
While generally idiopathic, choroidal folds can be seen in a wide variety of ophthalmic and systemic conditions, ranging from hyperopia to infectious, neoplastic, inflammatory, infiltrative, or immunologic disorders; thus, distinguishing the underlying condition is essential.2,3
Herein, we present a case of choroidal folds in a middle-aged man, along with the clinical appearance, etiology, pathophysiology, differential diagnosis, and management of this condition.
CASE REPORT
A 63-year-old White man presented for his annual diabetic eye examination. His past medical history included hyperlipidemia and type 2 diabetes with a hemoglobin A1c of 7%. His past ocular history was unremarkable except for new-onset and progressive hyperopia (+1.00 OD, +1.75+0.5 x 040 OS).
On examination, his BCVA was 20/20-3 OD and 20/30-2 OS. The anterior segment of each eye was unremarkable on slit-lamp evaluation except for 1+ nuclear sclerotic cataract in each eye.
The fundus examination of the right eye revealed no vitreous cell, normal optic nerve with a cup-to-disc ratio of 0.2, normal retinal vessels, and faint choroidal folds (Figure 1A). The left eye was similar, with the addition of RPE changes located peripherally and nasally to the optic nerve (Figure 1B). B-scan ultrasonography was normal, with no choroidal thickening or fluid. Fundus autofluorescence showed linear hyperreflective streaks compatible with the pattern seen on fundus photography (Figure 2).

Figure 1. Color fundus photos of the right (A) and left (B) eye showed a linear pattern of pigmentary changes in the posterior pole.
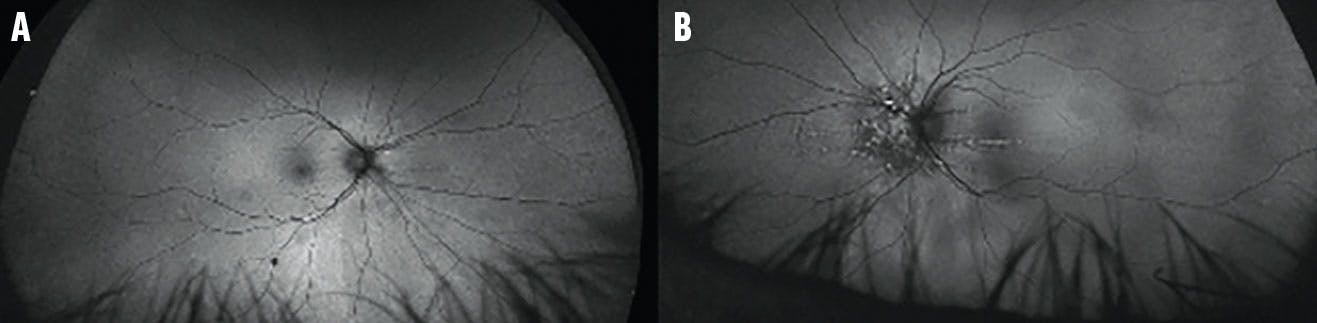
Figure 2. Fundus autofluorescence of the right (A) and left (B) eye showed linear hyperreflective streaks compatible with the pattern noted on fundus photography.
OCT revealed characteristic undulations of the RPE consistent with choroidal folds (Figure 3). Fluorescein angiography (FA) highlighted the choroidal folds in each eye (Figure 4). ICG did not reveal any choroidal masses (Figure 5), and a CT of the orbit was within normal limits. Blood workup was unremarkable.

Figure 3. OCT of the right (A) and left (B) eye showed the characteristic undulations of the RPE consistent with choroidal folds with a nearly flat retinal surface and a variable retinal thickness.
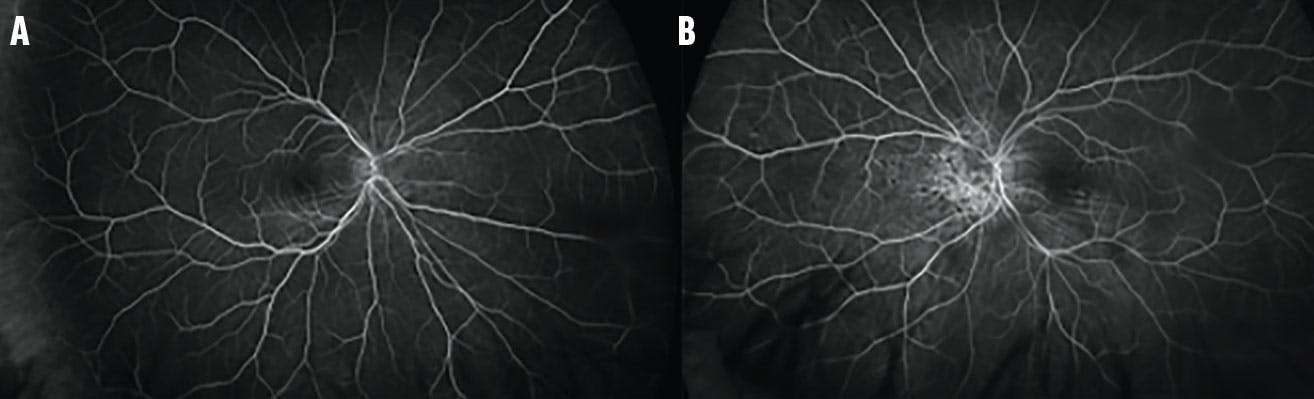
Figure 4. FA showed an increased stippled hyperfluorescence with late staining corresponding to an area of pigmentary disturbance in the right (A) and, more prominently, the left (B) eye.
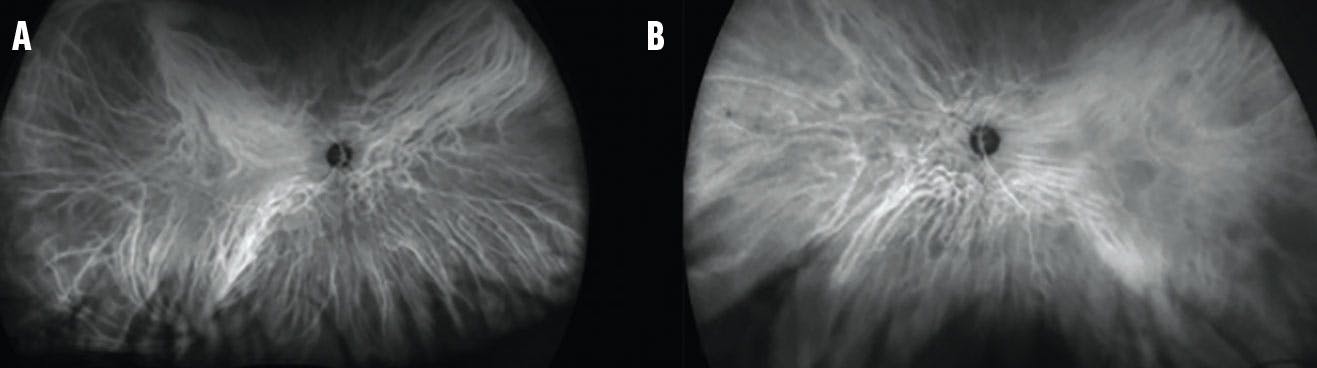
Figure 5. ICG revealed no choroidal lesions or neovascularization in either the right (A) or left (B) eye.
After ruling out all potential major causes of the choroidal folds, our patient was diagnosed with bilateral idiopathic choroidal folds. He has been followed for the past few years through annual diabetic eye examinations, with stable pathology.
POTENTIAL CAUSES
Choroidal folds can develop due to shrinkage of the inner sclera, infiltration or effusion into the choroid, or mechanical deformities. They may also occur due to various ocular conditions, such as hyperopia, hypotony, disciform scars, posterior scleritis, scleral buckling surgery, orbital tumors, uveal effusion, and AMD. Systemic conditions, including sinusitis, enlarged lacrimal gland caused by dacryoadenitis, idiopathic intracranial hypertension, thyroid eye disease (often in visually threatening forms), and complications of prolonged space flight among astronauts are other potential causes of choroidal folds.3,5-8
In a case series of 59 eyes with choroidal folds, Cangemi et al reported hyperopia as the most common cause; 17% of cases were idiopathic.1 Leahey et al likewise conducted a case series of 78 eyes with choroidal folds and revealed that AMD and hyperopia, followed by idiopathic, were the most common etiologies.9
Olsen et al proposed that with advances in diagnostic testing, the number of patients diagnosed with idiopathic choroidal folds will be reduced.3 They reported six cases of idiopathic choroidal folds in a series of 40 patients with choroidal folds, where five had a history of autoimmune disease. Thus, mild and undiagnosed ocular inflammatory conditions may contribute to the formation of choroidal folds.3
DIAGNOSTIC PEARLS
Multimodal imaging can help characterize the pathology and, in combination with a systemic workup, distinguish the etiology of choroidal folds.3 B-scan ultrasonography should be performed if posterior scleritis is suspected.
OCT is the most specific imaging tool to detect and distinguish idiopathic choroidal folds from other etiologies, such as epiretinal membrane.10 FA is the most sensitive imaging modality, where choroidal folds usually present as alternating hypofluorescent and hyperfluorescent bands due to rarefaction and redundancy of the RPE at the folds’ peaks and pits, respectively.10 On ICG, a delay in the choroidal vessel filling has been reported in cases of choroidal folds caused by orbital tumor.10
To exclude extraocular causes, neuroimaging and lumbar puncture in cases of papilledema should be considered. In aged eyes, loss of elasticity in the choroid-Bruch membrane complex can lead to thickening, fragility, and calcification of the Bruch membrane. Fracture of the Bruch membrane due to the strain created by choroidal folds in an ischemic environment may lead to CNV.11 Olsen et al reported four cases of CNV related to choroidal folds; each patient received anti-VEGF therapy, but none showed visual improvement. Visual deterioration was reported in two cases post anti-VEGF injections.3
Although our patient was diagnosed with idiopathic choroidal folds, there are some reports of choroidal folds among patients with diabetes with and without diabetic retinopathy.12,13 We postulate that the thick choroid and alterations in the choroidal sublayers secondary to diabetes may be the cause of the choroidal folds noted in our patient.
BEWARE OF A MASQUERADER
Choroidal folds can be mislabeled as retinal folds; however, retinal folds are much finer, are not limited to the posterior pole, and can present in various alignments. They are frequently seen in cases of epiretinal membranes after intraocular surgery, particularly vitreoretinal procedures. FA and ICG are helpful modalities in distinguishing retinal folds, which present as periodic surface undulations that do not involve the deep layers of the choroid.3,4 In addition, linear signal decreases in choriocapillaris perfusion captured on OCT angiography may be associated with choroidal folds.10,14
DON’T NEGLECT APPROPRIATE TESTING
While choroidal folds require no intervention, distinguishing and managing any underlying pathology is essential. Failure to identify an association between choroidal folds and tumors or CNV can lead to severe, irreversible vision loss. In addition, chronic choroidal folds with macular involvement can lead to maculopathy; thus, long-term follow-up with multimodal imaging and appropriate treatment is recommended.3 A step-by-step algorithm of management can aid in clinical decision making when you encounter choroidal folds (Figure 6).
1. Cangemi FE, Trempe CL, Walsh JB. Choroidal folds. Am J Ophthalmol. 1978;86(3):380-387.
2. Musetti D, Nicolò M, Bagnis A, Traverso CE. Chorioretinal folds: associated disorders and a related maculopathy. Am J Ophthalmol. 2014;158(2):409.
3. Olsen TW, Palejwala NV, Lee LB, Bergstrom CS, Yeh S. Chorioretinal folds: associated disorders and a related maculopathy. Am J Ophthalmol. 2014;157(5):1038-1047.
4. Friberg TR. The etiology of choroidal folds. A biomechanical explanation. Graefes Arch Clin Exp Ophthalmol. 1989;227(5):459-464.
5. Bagnis A, Cutolo CA, Corallo G, Musetti D, Nicolò M, Traverso CE. Chorioretinal folds: a proposed diagnostic algorithm. Int Ophthalmol. 2019;39(11):2667-2673.
6. Tran AQ, Zhang-Nunes SX, Cahill K, et al. Thyroid eye disease with choroidal folds. Orbit. 2021;40(3):206-214.
7. Cohen SY, Ducos de Lahitte G, Gaudric A, Mrejen S. Chorioretinal folds in patients with centeral serous chorioretinopathy. Retin Cases Brief Rep. 2022;16(2):242-245.
8. Ferguson CR, Pardon LP, Laurie SS, et al. Incidence and progression of chorioretinal folds during long-duration spaceflight. JAMA Ophthalmol. 2023;141(2):168-175.
9. Leahey AB, Brucker AJ, Wyszynski RE, Shaman P. Chorioretinal folds. A comparison of unilateral and bilateral cases. Arch Ophthalmol. 1993;111(3):357-359.
10. Grosso D, Borrelli E, Sacconi R, Bandello F, Querques G. Recognition, diagnosis and treatment of chorioretinal folds: current perspectives. Clin Ophthalmol. 2020;14:3403-3409.
11. Friberg TR, Lace JW. A comparison of the elastic properties of human choroid and sclera. Exp Eye Res. 1988;47(3):429-436.
12. Juan YB, Govindasamy G, Nadarajah G, Samsuddin AB. Case of idiopathic chorioretinal folds. Ann Clin Case Rep. 2021;6:1969.
13. Fagúndez Vargas MA, Jiménez Parras R, Bermúdez Uría L. Choroidal folds in diabetic retinopathy [article in Spanish]. Arch Soc Esp Oftalmol. 2000;75(12):797-802.
14. Comacchio F, Zorzi G, Sacconi R, Laesser R, Pichler A. Increased choroidal thickness in a patient with acquired hyperopia and choroidal folds syndrome. Am J Ophthalmol Case Rep. 2023;29:101803.












_1773249222.png?auto=compress,format&w=75)












