AT A GLANCE
- For some patients, faricimab (Vabysmo, Genentech/Roche) and high-dose aflibercept (Eylea HD, Regeneron) have the potential to provide greater durability than first-generation agents for wet AMD.
- Consider using a newer therapeutic for patients with wet AMD who have demonstrated a suboptimal treatment response or failed extension of their treatment interval.
- When contemplating a switch to a second-generation therapy, consider whether the patient is treatment-naïve, if they are sensitive to IOP increases post-injection, and if they have a history of ocular inflammation.
- Some people’s disease can be quite recalcitrant to treatment and will still require frequent treatment.
With the advent of next-generation therapies for the treatment of wet AMD, clinicians have much to consider when deciding the proper treatment approach for patients with neovascularization. I (L.R.) spoke with retina specialists at Wills Eye Hospital about their various approaches to treating wet AMD. Here, they discuss when they prefer to continue with current therapy and when they decide to switch patients to faricimab (Vabysmo, Genentech/Roche) or high-dose aflibercept (Eylea HD, Regeneron). They also explain when they might consider a port delivery system (PDS) with ranibizumab (Susvimo, Genentech/Roche), when it becomes commercially available.
THOUGHTS FROM CARL D. REGILLO, MD
Second-generation anti-VEGF agents like faricimab and high-dose aflibercept have shown, to some degree, greater durability than first-generation agents for wet AMD. They are potentially useful for any patient with wet AMD, both newly diagnosed and previously treated. In real-world practice, however, most commercial payors require starting new patients on a first-generation agent. This often involves multiple treatments of two different first-generation agents before approving coverage for one of the two second-generation agents.
For established, previously treated patients, if they are doing well on a given anti-VEGF agent and being dosed relatively infrequently (eg, every 10 to 12 weeks), I will offer them the option to switch and potentially extend out longer. Most of the time, patients prefer to stay on their existing treatment, and in general, I am relatively reluctant to extend patients beyond 12 weeks.
However, for patients on an older agent who are unable to have their injections extended beyond 8 weeks, I will discuss switching and often make that switch to potentially decrease their treatment burden (Case 1). Typically, I can extend a frequently treated patient’s interval by 1 to 2 weeks. Furthermore, even if I can’t extend the treatment interval after switching, I can often get better exudative control in patients with suboptimal disease control (ie, persistent exudation) on an older agent.
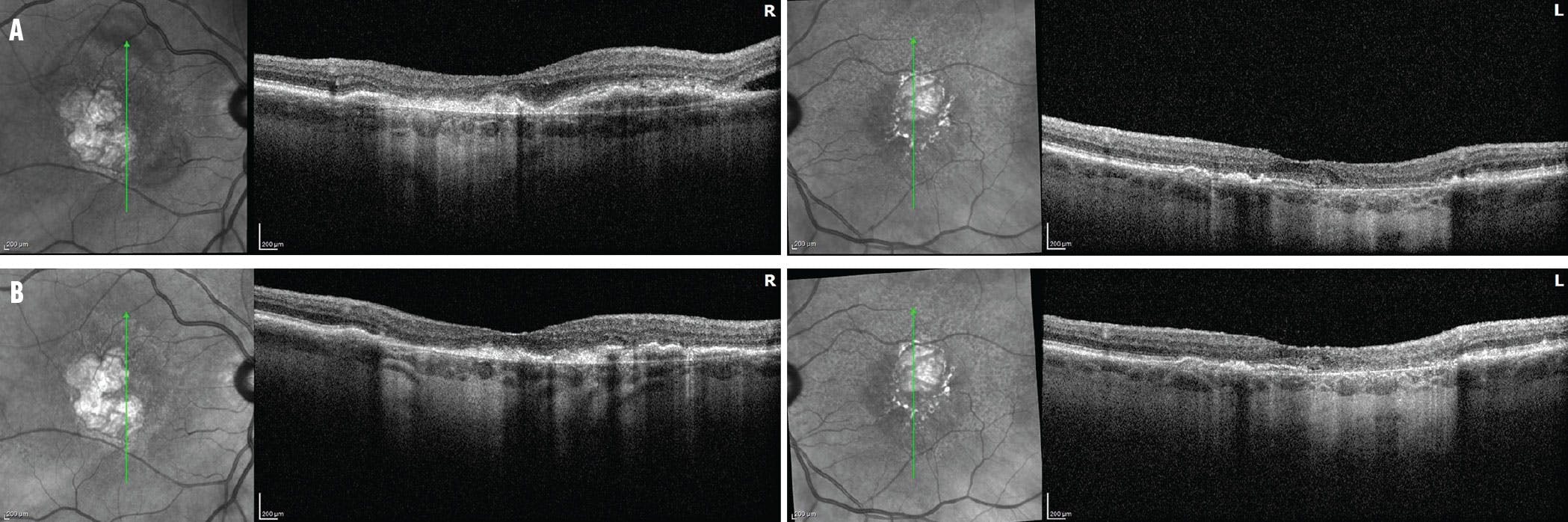
Case 1. An 83-year-old man with a history of wet AMD and geographic atrophy in each eye was being treated with 2 mg aflibercept every 7 weeks with worsening exudation (A). His VA was 20/200 OD and 20/100 OS. The patient switched to faricimab in each eye with a better drying effect and was extended to 8 weeks (B). His current VA is 20/100 OD and 20/50 OS. The patient is not receiving treatment for geographic atrophy at this time.
Regarding the PDS, I look forward to the device being reintroduced into the commercial setting. I had a favorable experience using it to treat wet AMD as a maintenance therapy in phase 2 and 3 clinical trials. Existing patients who are not able to be extended beyond 8 weeks, even after trying a second-generation agent, may benefit from the greater durability that comes with the sustained-release approach of the PDS. I would offer this device as an option in such patients after reviewing, in detail, the risks and benefits of having it implanted.
TREATMENT DELIBERATIONS FOR SONIA MEHTA, MD
I consider new wet AMD therapies such as faricimab or high-dose aflibercept for patients with persistent disease or new-onset wet AMD, if insurances allow them. Specifically, if the patient is doing well on 2 mg aflibercept (Eylea, Regeneron) but fails extension, I consider switching to high-dose aflibercept (Case 2). After the third injection of high-dose aflibercept, labeling allows for repeat injections every 7 to 8 weeks or further, but not sooner.
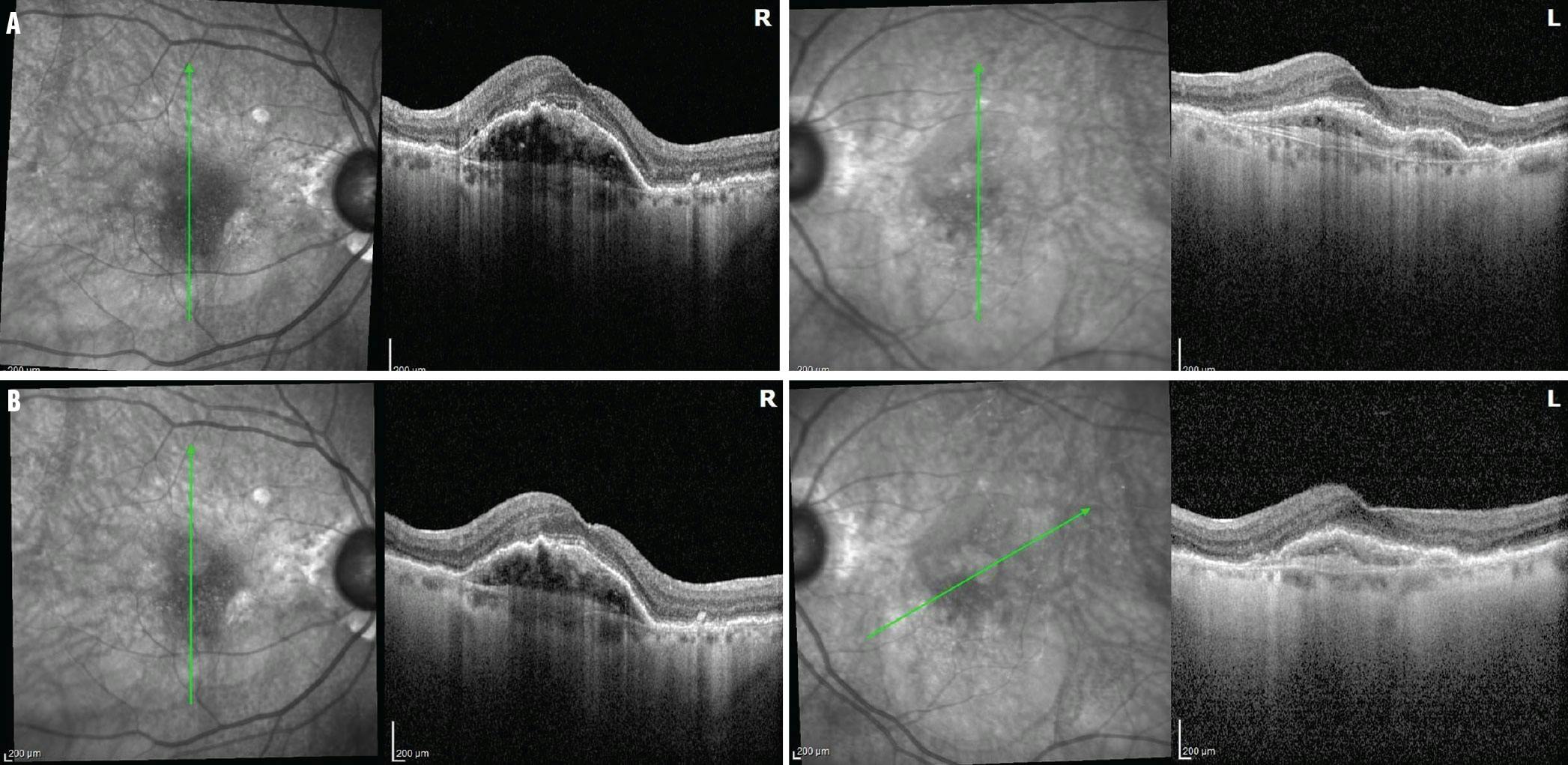
Case 2. A 90-year-old woman with wet AMD in each eye was being treated with intravitreal faricimab every 8 weeks with persistent exudation in the right eye (A). Her VA was 20/60 OD and 20/50 OS. The patient switched to high-dose aflibercept in the right eye and continued on faricimab in the left (B). Her VA improved to 20/30 OD and 20/50 OS.
If I am using a second-generation medication and need more flexibility in the treatment interval, faricimab would be a good option. Also, if a patient has issues with elevated IOP post injection, ocular hypertension, or advanced glaucoma with severe cupping, I may favor faricimab.
I would consider the PDS for patients who are having a great response to ranibizumab (Lucentis, Regeneron) and would like to decrease their injection burden. I do a careful preoperative assessment and evaluate the conjunctiva and sclera. However, there is a risk for infection with a long-term surgical implant. Gene therapies also look promising as a modality to reduce injection burden in the future.
Still, some patients have disease that is well controlled with first-generation medications at 2- to 3-month treatment intervals. Switching agents may require decreasing treatment intervals initially or going through a step therapy program prior to getting insurance authorization for the second-generation medication.
SWITCHING CONSIDERATIONS FOR SUNIR J. GARG, MD
Although I have some patients who are treatment-naïve, most of the patients I treat with faricimab and high-dose aflibercept have been refractory to ranibizumab or 2 mg aflibercept. These refractory patients were not part of the clinical studies. I have also been using these newer drugs for patients who respond well to first-generation drugs but must come in every 4 to 6 weeks. Because faricimab was FDA-approved before high-dose aflibercept, more insurance carriers in our area have a medical policy for it (often only after we have exhausted a step therapy ladder).
When I start using faricimab, I generally do three monthly treatments and then slowly extend. I have had good results in getting patients out further than they were before. Because these patients have been previously treated, however, I’m not able to extend them out as much as was seen in the studies. I am also using high-dose aflibercept, and I find that it’s an easy choice for patients who are currently receiving 2 mg aflibercept. I have fewer patients on it due to its more recent approval and more variable insurance coverage in our area.
I caution patients, however, that the disease can be quite recalcitrant to treatment at times, and no matter what we do, some patients only experience a benefit from frequent treatment with anti-VEGF medications. In addition, some patients still benefit from steroid treatments and photodynamic therapy.
However, in a subset of patients who were stuck at 4-week intervals for years, I have been able to get them as far out as every 10 weeks. Many of these people are understandably hesitant to push their intervals out too far, but the longer time out of the office is welcome.
REFLECTIONS FROM ALLEN CHIANG, MD
Since we live in the age of step therapy, I usually consider the newer wet AMD therapies for patients who have either demonstrated a suboptimal treatment response (eg, persistent exudation despite monthly injections) or failed extension of their treatment interval (Case 3). Since high-dose aflibercept is newer than faricimab, I don’t have as much real-world experience with it yet, but I anticipate that its real-world performance will be similar.

Case 3. A 70-year-old man with wet AMD in the left eye was receiving treatment every 4 weeks with 2 mg aflibercept with residual exudation (A). The patient was transitioned to faricimab and extended to 9 weeks with a better drying effect (B).
To date, my personal experience with faricimab has been more modest than the clinical trials. Admittedly, my approach with these new agents has been to start with my most difficult patients. The PDS has some amazing research and development behind it, but as a surgically implanted device, it will always pose some unique considerations in an elderly wet AMD patient population. In addition, the voluntary recall gives me pause. The PDS will likely retain a small niche within our treatment armamentarium, but I don’t see it moving beyond that, especially with the increasing durability of our injectable options.
TALKING POINTS FROM MICHAEL A. KLUFAS, MD
When choosing the right agent to treat wet AMD, there are some points to consider. If a patient is treatment-naïve versus treatment-experienced, the outcomes may vary. Because the phase 3 trials only included treatment-naïve wet AMD patients, we don’t know if we will see the same effect in patients who were previously treated.
I have implanted both the clinical trial PDS and the commercial PDS prior to the recall. I bring this option up first, as I do believe the ideal candidate for this type of therapy is a patient who loathes the intravitreal injection process, especially when they report being worn out or the day being lost after an intravitreal injection. Another good candidate for the PDS is a patient who requires anti-VEGF therapy every 2 to 3 weeks to stay dry; this is rare, but the continuous delivery provided by the PDS may address this high anti-VEGF burden.
If I am considering initiating faricimab or high-dose aflibercept, there are several features I consider when making the choice. Because high-dose aflibercept is a slightly higher volume (.07 cc), I may consider faricimab over high-dose aflibercept for patients with severe glaucoma or high sensitivity to intravitreal injection volumes greater than .05 cc. In addition, any biologic can produce intraocular inflammation, though usually (and luckily) not occlusive retinal vasculitis. For instance, if a patient has tolerated 2 mg aflibercept well, but I am not achieving the durability I would like, I might consider switching to high-dose aflibercept because it is the same compound. There have been some cases of intraocular inflammation reported with faricimab, and if a patient has a history of recurrent inflammation, I might consider a legacy product such as ranibizumab, which has very low rates of intraocular inflammation.
Finally, it is important to discuss with the patient any need to switch. Almost daily, I have a patient who is on 2 mg aflibercept every 6 or 8 weeks, and I discuss the option of switching to high-dose aflibercept to try to achieve a longer treatment interval. Many of my wet AMD patients, especially those doing well on 2 mg aflibercept, are not eager to switch to a different therapy.
On the other hand, for patients who have persistent exudation even with monthly dosing, I worry that persistent exudation may adversely affect long-term vision, and I suggest changing to faricimab or high-dose aflibercept (Case 4). In my experience, some patients respond to certain agents better than others. My hypothesis is that eyes that are VEGF-sensitive or have high VEGF load may do better on aflibercept, whereas eyes that have vascular instability that is mediated by more than VEGF may do better with faricimab and angiopoetin-2 inhibition.

Case 4. An 80-year-old man with wet AMD in the left eye was receiving intravitreal 2 mg aflibercept every 4 weeks with persistent exudation (A). His VA was 20/60 OS. The patient switched to high-dose aflibercept in the left eye and has been extended to 10 weeks (B). His VA improved to 20/40 OS and remains stable.
Importantly, one of the major factors determining treatment choice is insurance plans. Some plans even have double-step therapies, which can limit the initial use of the physician-selected products.
FINAL TIPS FROM AJAY E. KURIYAN, MD
At this point, I am using newer wet AMD therapies for patients who cannot be extended on their current agents due to persistent fluid. We have more experience and flexibility for dosing with faricimab. High-dose aflibercept, however, has the limitation of not being approved for use more frequently than every 7 weeks after the third loading dose. This is an important consideration for patients who may not be extended for 7 or more weeks.
I have yet to switch patients between farcimab and high-dose aflibercept, but I have seen examples of patients who have better anatomic outcomes after switching between these two agents. This likely means that there are some patient factors that affect treatment response. Although I discuss the PDS option with patients who are particularly unhappy about injections, my patients have preferred to continue with injections over a more invasive option.















_1773249222.png?auto=compress,format&w=75)












