Over the past few years, our understanding of inherited vitreoretinopathies and vitreoretinal degenerations has significantly advanced, particularly when it comes to the identification of causative genes.1 Vitreoretinal degenerations are a group of retinal disorders marked by early-onset cataracts, liquefaction of the vitreous humor, variable retinal degeneration, and abnormalities at the interface between the vitreous and the retina. These abnormalities increase traction on the retina, leading to retinal detachment (RD). Most of these conditions, including snowflake vitreoretinal degeneration (SVD), are autosomal dominant diseases with variable expressivity and nearly complete penetrance of vitreoretinal degeneration.2,3 It is important to consider SVD in the differential diagnosis of patients with fibrillar vitreous anomalies.4,5
Here, we discuss a patient diagnosed with SVD with a macular hole, RD, and complications in the left eye who followed up with complications in the right eye.6
THE CASE
A healthy 18-year-old female patient presented with a history of amblyopia, high myopia, and increased blurry vision in the right eye for the past 2 years. Her BCVA was 20/200 OD and 20/25 OS. Anterior segment examination was unremarkable. Indirect fundoscopy revealed flattened, small optic nerves, cystic changes in the macula and retinal periphery of each eye, and a full-thickness macular hole (FTMH) in the right eye. Fluorescein angiography demonstrated peripheral straightening of vessels and no retinal vascular leakage. OCT confirmed the flat macular schisis and FTMH in the right eye. OCT also showed macular schisis and vitreomacular traction in the left eye. As FTMHs are rare in the pediatric population, further investigation was necessary. Genetic studies disclosed a heterozygous pathogenic variant in the KCNJ13 gene, consistent with SVD. The patient elected for observation of the FTMH.
The patient developed a FTMH in the left eye 17 months later, followed by a rhegmatogenous RD (RRD) with a VA of light perception requiring two surgical repairs, including a scleral buckle, external drainage, cryopexy, and 100% C3F8 gas for the first attempt and an RRD/FTMH repair with pars plana vitrectomy (PPV), membrane and internal limiting membrane peeling, PFO, endolaser, and silicone oil injection (1,000 centistokes) for the second. One month later, there was an RD from the FTMH in the right eye with a VA of counting fingers (Figure 1A and B).

Figure 1. Eighteen months after surgical intervention for the left eye, fundus (A) and OCT (B) imaging of the right eye reveal an RD through the macular hole. At postoperative day 30 after PPV in the right eye (C and D), the retina is attached. At postoperative month 3 (E and F), the retina is redetached with accompanying proliferative vitreoretinopathy. Courtesy of Giselle de Oliveira.
PPV was pursued in the right eye to repair the RRD with membrane peeling, PFO, endolaser, and silicone oil injection (1,000 centistokes). Intraoperatively, the vitreous was noted to be adherent and fibrillar. The retina was attached on postoperative day 30 (Figure 1C and D), but by postoperative month 3 (Figure 1E and F), the patient developed a redetachment with proliferative vitreoretinopathy. She underwent another RRD repair with PPV, membrane peeling, PFO, endolaser, and silicone oil injection (5,000 centistokes). The retina remained attached in each eye, but the FTMHs persisted in each eye with a VA of 20/200 OD and 20/250 OS at postoperative month 6 (Figure 2).
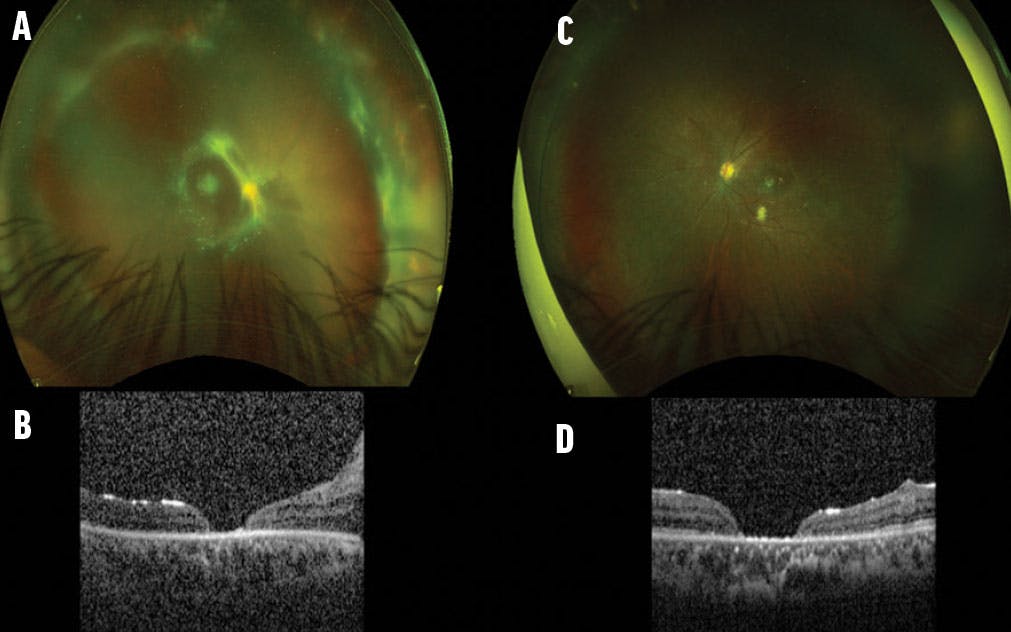
Figure 2. Fundus and OCT imaging of the right (A and B) and left (C and D) eyes at postoperative month 6 after the final intervention in the right eye. . Courtesy of Giselle de Oliveira.
DISCUSSION
SVD is a rare genetic condition that significantly affects the retina. This disorder is characterized by distinctive crystalline deposits in the vitreous and retina, resembling snowflakes that give the condition its name.7 These deposits are indicative of the underlying degenerative processes that affect retinal structure and function. While there were no corneal guttata, cataracts, or perivascular sheathing in this case, salient features of SVD included a fibrillar vitreous structure, crystalline retinopathy, and flattened optic nerves.
The primary challenges in managing SVD are its genetic basis and rarity. As an inherited condition, it often requires genetic counseling and family screening to understand the inheritance patterns and identify at-risk individuals. The rarity means there is limited data available, making diagnosis and treatment particularly challenging, especially with the oftentimes subtle and variable initial presentation.
Although pathological myopia and RRD have been described in SVD,2,5,8 retinoschisis and FTMHs have not been previously associated with this syndrome. No participants in the Hirose et al study were reported to have a macular hole, a finding that is often related to blunt trauma in pediatric patients.2,5,9 While abnormal vitreous traction has been described in myopic degeneration, it may also be associated with SVD.10,11 We speculate that the fibrillar vitreous structure is anchored abnormally to the retina in SVD, contributing to vitreomacular traction, FTMHs, and RRD.
RD has been reported in up to 21% of SVD patients.5 Compared with other retinal degenerations, such as Stickler syndrome, SVD patients have a much higher rate of RD (65%).12 Pediatric RDs are often more complex than those in adults due to late presentation, associated ocular anomalies, pathology in the fellow eye, and a tendency for bilateral occurrence.13 These detachments frequently require multiple surgeries to achieve anatomical success, with each procedure carrying risks such as infection, cataract formation, inflammation, and additional retinal damage.14
Pediatric patients with SVD require lifelong monitoring to manage the recurrent and progressive nature of the disease. Decision making in pediatric vitrectomies must be individualized and consider the patient’s overall development, progression of the disease, and psychosocial effect on the patient and their family. Multidisciplinary care teams, including retina specialists, pediatric ophthalmologists, and geneticists, play a vital role in managing these young patients and providing comprehensive care.
PROCEED WITH CAUTION
SVD presents significant challenges due to its rarity and genetic basis. The retinal complications of macular holes and RD necessitate timely and often repeated surgical intervention, which can be particularly challenging in young patients. Pediatric vitrectomies require long-term follow-up and careful, individualized decision making to preserve vision and quality of life for these patients.
1. Edwards AO. Clinical features of the congenital vitreoretinopathies. Eye (Lond). 2008;22(10):1233-1242.
2. Hirose T, Lee KY, Schepens CL. Snowflake degeneration in hereditary vitreoretinal degeneration. Am J Ophthalmol. 1974;77:143-153.
3. Richards AJ, Scott JD, Snead MP. Molecular genetics of rhegmatogenous retinal detachment. Eye. 2002;16:388-392.
4. Jiao X, Ritter R III, Hejtmancik JF, Edwards AO. Genetic linkage of snowflake vitreoretinal degeneration to chromosome 2q36. Invest Ophthalmol Vis Sci. 2004;45(12):4498-4503.
5. Lee MM, Ritter R III, Hirose T, Vu CD, Edwards AO. Snowflake vitreoretinal degeneration: follow-up of the original family. Ophthalmology. 2003;110(12):2418-2426.
6. Ashkenazy N, Sengillo JD, Iyer PG, Negron CI, Yannuzzi NA, Berrocal AM. Phenotypic expansion of KCNJ13-associated snowflake vitreoretinal degeneration. Ophthalmic Genet. 2023;44(5):505-508.
7. Hejtmancik JF, Jiao X, Li A, et al. Mutations in KCNJ13 cause autosomal-dominant snowflake vitreoretinal degeneration. Am J Hum Genet. 2008;82(1):174-180.
8. Edwards A, Robertson JE. Hereditary vitreoretinal degenerations. In: Ryan S, Hinton D, Schachat A Wilkenson P, eds. Retina. 4th ed. Elsevier; 2006:519-538.
9. Kothari N, Read SP, Baumal CR, et al. A multicenter study of pediatric macular holes: surgical outcomes with microincisional vitrectomy surgery. J Vitreoretin Dis. 2019;4(1):22-27.
10. Kobayashi H, Kobayashi K, Okinami S. Macular hole and myopic refraction. Br J Ophthalmol. 2002;86(11):1269-1273.
11. Tey K, Wong QY, Dan YS, et al. Association of aberrant posterior vitreous detachment and pathologic tractional forces with myopic macular degeneration. Invest Ophthalmol Vis Sci. 2021;62(7):7.
12. Ang A, Poulson AV, Goodburn SF, Richards AJ, Scott JD, Snead MP. Retinal detachment and prophylaxis in type 1 Stickler syndrome. Ophthalmology. 2008;115(1):164-168.
13. Soliman MM, Macky TA. Pediatric rhegmatogenous retinal detachment. Int Ophthalmol Clin. 2011;51(1):147-171.
14. Fivgas GD, Capone A Jr. Pediatric rhegmatogenous retinal detachment. Retina. 2001;21(2):101-106.












_1773249222.png?auto=compress,format&w=75)












