In recent years, significant advances in the treatment of geographic atrophy (GA) have been made with the FDA approval of pegcetacoplan (Syfovre, Apellis) and avacincaptad pegol (Izervay, Astellas). While these therapies—both of which target the complement system to slow GA progression—offer promising benefits, they also carry risks, including the potential development of wet AMD.
The decision to treat patients remains complex, requiring careful consideration of individual patient factors and an in-depth patient discussion of potential risks and benefits. The following cases depict patients who developed wet AMD within 10 months of commencing anti-complement therapy for GA, highlighting the need for careful screening and monitoring of patients being started on these therapies.
CASE 1
An 83-year-old man presented with a 3-year history of progressive visual decline in his left eye. His past ocular history was notable for longstanding poor vision in the right eye with BCVA of counting fingers secondary to macular neovascularization (MNV). The left eye, historically with a BCVA of 20/30, had deteriorated to 20/70 at presentation. The anterior segment examination was unremarkable. Indirect ophthalmoscopy revealed retinal pigment epithelium (RPE) changes in each eye. Imaging studies, including red-free imaging, OCT, and fundus autofluorescence (FAF), demonstrated significant GA progression in the left eye (Figure 1). Given the progression, intravitreal pegcetacoplan therapy was initiated in the left eye and administered every 8 weeks. Eight weeks after the fourth injection, the patient’s VA worsened to 20/200 OS with findings of subretinal hemorrhage and pigment epithelial detachment (PED; Figure 2). There was no evidence of exudative changes at the previous visit. Pegcetacoplan was discontinued, and anti-VEGF therapy was started. While the PED and hemorrhage resolved, the BCVA remained at 20/200 with GA. The patient declined further anti-complement therapy.

Figure 1. FAF shows significant progression of the GA lesion in the left eye from baseline (A) to 9 months (B). Note the large area of RPE loss leading to hypoautofluorescence with sickened RPE along the edge of the atrophic area seen as hyperautofluorescence.
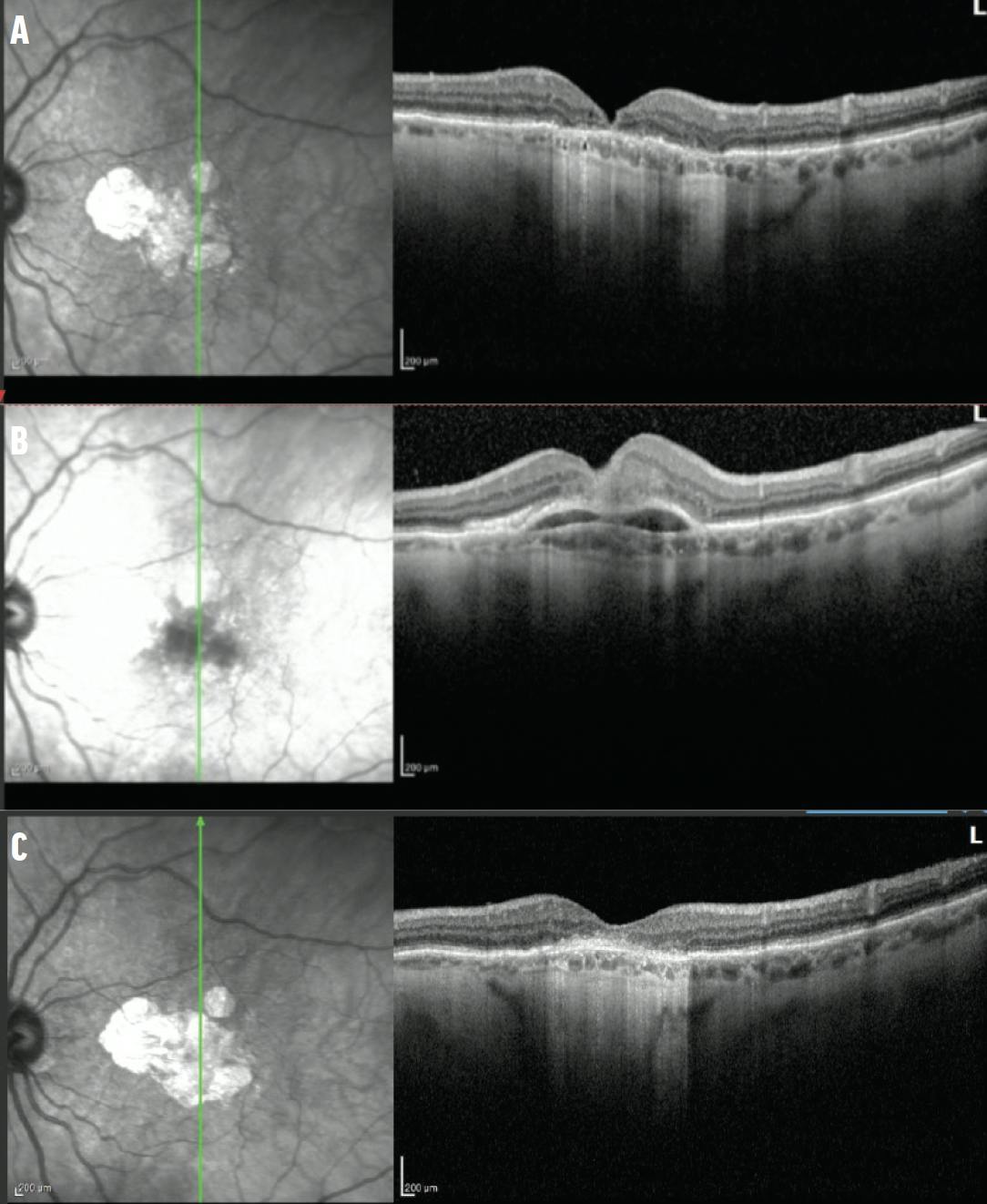
Figure 2. OCT imaging documents the transformation from dry (A) to wet (B) AMD 8 weeks after the fourth injection of pegcetacoplan. Note the fibrovascular PED with associated subretinal hemorrhage. After anti-VEGF therapy, OCT imaging shows resolution of the hemorrhage and PED (C).
CASE 2
A 78-year-old woman presented with a history of dry AMD and a gradual decline in visual acuity due to GA progression in the left eye. Intravitreal avacincaptad pegol was initiated. After the fourth injection, the patient’s VA declined from 20/50 to 20/100. Clinical examination and OCT revealed new intraretinal fluid and subretinal hyperreflective material, indicating conversion to wet AMD (Figure 3). Intravitreal bevacizumab (Avastin, Genentech/Roche) was administered; however, subsequent follow-up showed worsening fluid. The treatment was then switched to 8 mg aflibercept (Eylea HD, Regeneron), which led to a reduction in intraretinal fluid. At the 4-month follow-up, VA had improved to 20/60. The plan is to continue both anti-VEGF and anti-complement treatment.

Figure 3. This patient with GA and new-onset wet AMD (A) experienced progression of intraretinal fluid despite treatment with intravitreal bevacizumab (B). OCT imaging shows improvement of the intraretinal fluid compared with baseline (C) after switching to intravitreal 8 mg aflibercept (D).
DISCUSSION
The emergence of complement inhibition therapy has introduced new tools for managing GA.1,2 However, these treatments carry a risk of conversion to wet AMD.1-6 These cases highlight the potential risk of conversion and the importance of monitoring and prompt intervention in patients undergoing GA therapy. They also emphasize the potential risk of irreversible vision loss following conversion to wet AMD.
In the OAKS and DERBY trials evaluating pegcetacoplan, the incidence of new-onset wet AMD over 24 months was 12.2% in monthly dosing, 6.7% in every-other-month dosing, and 3.1% in the pooled sham arms.6 The majority of the MNV cases identified on fluorescein angiography were occult. Similarly, 24-month data from the GATHER2 trial assessing monthly 2 mg avacincaptad pegol reported choroidal neovascularization rates of 12% compared with 9% in the sham arm.5
Data from OAKS and DERBY showed that when pegcetacoplan was administered with anti-VEGF treatment, no unexpected outcomes were observed (the median change in BCVA from the study visit preceding wet AMD conversion to month 24 was -5.0 letters in those receiving pegcetacoplan monthly, -6.0 letters in those receiving pegcetacoplan every other month, and -5.0 letters in those receiving sham).1 These findings appear consistent with real-world data presented at the 2025 Macula Society meeting, in which patients maintained good vision despite wet AMD conversion.3
Data from GATHER1 and GATHER2 also demonstrated no significant difference in BCVA change from baseline to months 12 and 18 between patients treated with avacincaptad pegol and those receiving sham with the development of MNV. Among patients who developed MNV, the mean change in BCVA from baseline to month 12 was -4.2 letters in both the avacincaptad pegol and sham groups. At month 18, the mean change was -9.6 letters in the avacincaptad pegol group and -10.3 letters in the sham group.5
The cases reported here were associated with rare events. In case 1, the patient’s vision remained at 20/200 despite resolution and treatment of exudative changes. It is important to counsel patients that, although rare, conversion to wet AMD during anti-complement therapy for GA can lead to irreversible vision loss, even with prompt treatment. In case 2, the patient’s vision improved from 20/100 to 20/60 after the treatment of MNV, highlighting the importance of early detection and treatment of MNV conversion.
Certain baseline characteristics have been associated with an increased likelihood of developing wet AMD during complement inhibition therapy. In the phase 2 FILLY trial, the presence of MNV in the fellow eye and the double-layer sign on OCT were identified as significant risk factors.4 Case 1 had a history of MNV in the fellow eye and, thus, had a high risk of conversion. In both cases, the patients developed a subtle double-layer sign during the initial injections prior to the conversion to wet AMD (Figure 4).

Figure 4. OCT imaging of the left eye of the patient in case 1 (A) shows early development of a double-layer sign with increasing subretinal hyperreflectivity (B, arrow) during the injections with complement inhibition therapy prior to wet AMD conversion. Similarly, the left eye of case 2 (C) shows early development of a double-layer sign with increasing subretinal hyperreflectivity (D, arrow) during the injections with complement inhibition therapy prior to wet AMD conversion.
DOCUMENT AND SHARE
As the use of complement inhibition for GA increases, it is imperative to document and share clinical outcomes to enhance our understanding of their efficacy and safety profiles. Regularly monitoring patients for early signs of conversion to wet AMD is critical. During the trials, imaging with OCT angiography was not the standard protocol; however, given the risks of MNV conversion, we now include OCT imaging for new patients. In our practice, we carefully review all raster lines of OCT scans every visit and perform OCT angiography at the initiation of therapy, every 6 months, and earlier if new symptoms are reported or if there is any suggestion of new clinical changes. Looking to the future, ongoing research into alternative treatments for GA may one day broaden our therapeutic options.
1. Heier JS, Lad EM, Holz FG, et al; OAKS and DERBY study investigators. Pegcetacoplan for the treatment of geographic atrophy secondary to age-related macular degeneration (OAKS and DERBY): two multicentre, randomised, double-masked, sham-controlled, phase 3 trials. Lancet. 2023;402(10411):1434-1448.
2. Patel SS, Lally DR, Hsu J, et al. Avacincaptad pegol for geographic atrophy secondary to age-related macular degeneration: 18-month findings from the GATHER1 trial. Eye (Lond). 2023;37(17):3551-3557. Erratum in: Eye (Lond). 2023;37(17):3705.
3. Rosenfeld P, Shen M, Berni A, et al. Real-world efficacy and safety of intravitreal pegcetacoplan for the treatment of geographic atrophy in AMD. Presented at Macular Society; February 12-15, 2025; Charlotte Harbor, Florida.
4. Wykoff CC, Rosenfeld PJ, Waheed NK, et al. Characterizing new-onset exudation in the randomized phase 2 FILLY trial of complement inhibitor pegcetacoplan for geographic atrophy. Ophthalmology. 2021;128(9):1325-1336.
5. Iveric Bio announces positive 24-month topline results from phase 3 study of IZERVAY (avacincaptad pegol intravitreal solution) for geographic atrophy [press release]. Astellas. September 18, 2023. Accessed April 3, 2025. www.astellas.com/en/news/28506
6. Ferrone P. 24-Month safety results from the OAKS and DERBY trials with further characterization of exudative age-related macular degeneration. Presented at: Macula Society; February 15-18, 2023; Miami.











_1773249222.png?auto=compress,format&w=75)












