Oveitis masquerade syndromes encompass a range of conditions that mimic the clinical presentation of uveitis but are caused by underlying pathologies such as malignancies, infections, or degenerative diseases.1,2 Neoplastic masqueraders, such as intraocular lymphoma, uveal melanoma, leukemia, and metastatic tumors, are among the most serious and challenging to diagnose due to the potential for systemic involvement and life-threatening progression. Such conditions account for about 2.5% of presumed uveitis patients in tertiary care settings.3 The incidence varies in terms of geographic location, access to health care, and presence of systemic malignancies. Early detection hinges on identifying atypical features, using advanced imaging modalities, and performing targeted diagnostic procedures such as vitreous biopsy.
INTRAOCULAR LYMPHOMA
Primary vitreoretinal lymphoma (PVRL) is most common in older adults, with a peak incidence between 50 and 70 years of age. It is a subset of primary central nervous system (CNS) lymphoma, which itself accounts for 1% to 2% of all non-Hodgkin lymphomas.4,5 Secondary intraocular lymphoma is more frequent in patients with systemic lymphoma and has no clear age predilection.
A high frequency of MYD88 and CD79B mutations with VRL has been observed.4 In addition, it is important to remember the following pearls:5
- At the time of presentation with VRL, CNS involvement is present in 16% to 34% of patients.
- Of patients with primary CNS lymphoma, 25% have VRL at presentation.
- Of patients with VRL, 50% to 90% develop a CNS and/or spinal cord disease within 16 to 24 months.
Recent recommendations suggest the following elements should be used to obtain a PVRL diagnosis:6
- Clinical History: Most patients with PVRL are older than 50 years of age and present with floaters and painless vision loss without redness or photophobia. The disease may manifest unilaterally, but PVRL is predominantly bilateral.
- Anterior Segment Findings: Keratic precipitates of various types may or may not be present in VRL. Scleritis, pseudohypopyon, and hyphema are uncommon, while anterior synechiae and iris depigmentation are almost never seen.
- Vitreous and Posterior Segment Findings: Massive cellular infiltration (vitreous sheets or clumps) with the absence of macular edema, snowbanking, or vitreous hemorrhage may be present. There are usually multifocal creamy/white lesions in the outer retina (Figure 1) with or without a leopard-spot appearance, and retinal pigment epithelial (RPE) atrophy or fibrosis.
- Ocular Imaging: Multimodal imaging, including fundus autofluorescence (Figure 2), OCT (Figure 3), fluorescein angiography, and contrast-enhanced MRI of the brain, is also important to investigate.
- Behavior With Systemic Steroids: Systemic steroids should not be used as a sole treatment for VRL due to their limited effectiveness and should be stopped at least 2 weeks prior to a scheduled vitrectomy for ocular fluid study.
- Ocular Fluid Diagnosis: In cases of suspected VRL, diagnostic vitrectomy may be performed after oral steroids have been discontinued for at least 2 weeks. Early undiluted vitreous samples should be obtained with a low cut rate, and diluted and undiluted vitreous should be evaluated as soon as possible. Cell morphology and cytokine and gene analysis of the ocular fluid can aid in the diagnosis; the IL-10:IL-6 ratio and MYD88 gene mutation are strongly recommended. Immunophenotype (ie, flow cytometry, immunocytology, and histochemistry) and polymerous chain reaction testing should also be performed, if possible.

Figure 1. Fundus images of the right eye of a 55-year-old man on presentation showed multifocal creamy/white lesions in the outer retina with leopard-spot and RPE atrophy typical of intraocular lymphoma. This patient underwent vitrectomy, which confirmed the diagnosis, and he developed brain lymphoma 14 months later.
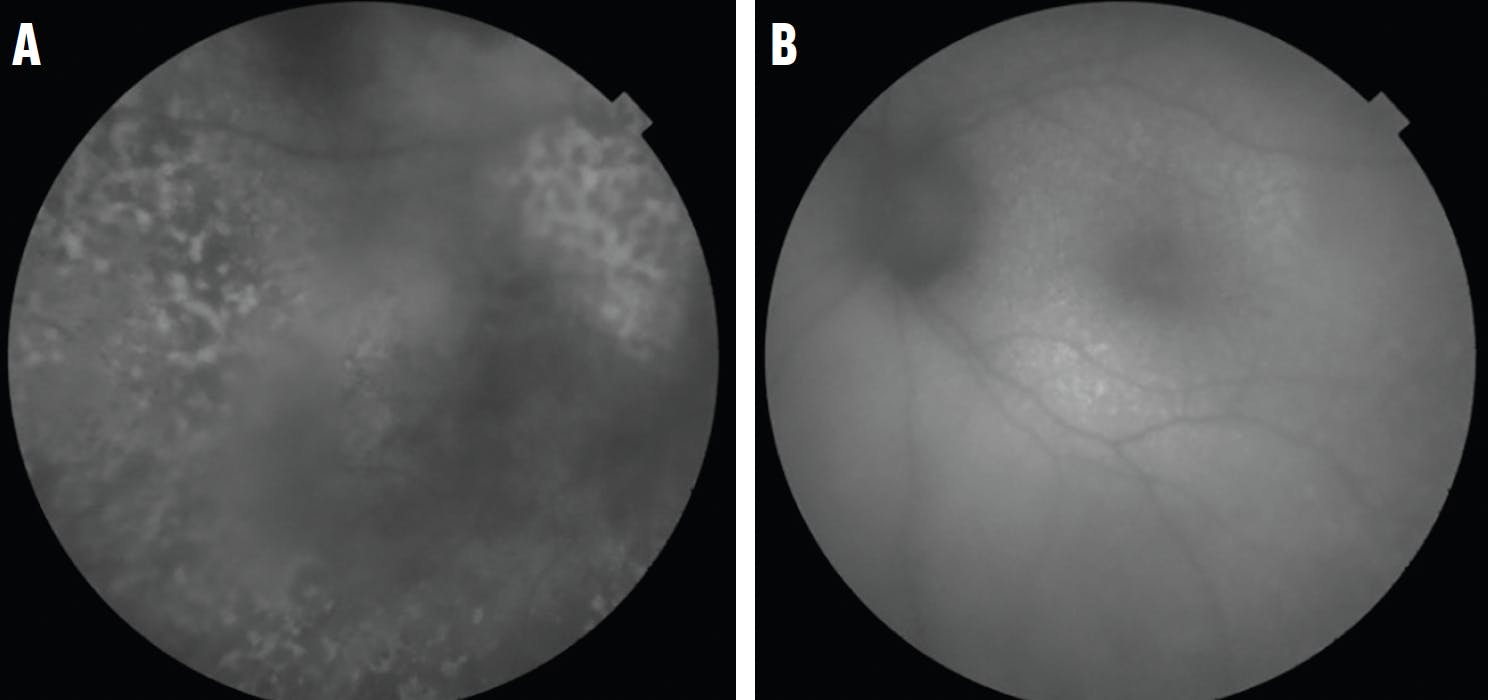
Figure 2. Fundus autofluorescence of the right (A) and left (B) eye of a 66-year-old woman showed a granular hyper- and hypoautofluorescence pattern indicative of VRL. This patient had a vitrectomy specimen labeled as “highly suspicious” for lymphoma and, indeed, developed brain lymphoma 2 months after presenting with ocular symptoms.

Figure 3. OCT of the patient in Figure 1 showed focal round lesions causing pigment epithelial detachment with RPE irregularities and subretinal fluid (A). In another case of PVRL in a 39-year-old man, whose ocular symptoms had been misdiagnosed as uvetitis, RPE thickening, subretinal fluid, and diffuse highly reflective subretinal infiltration were observed (B).
The Interleukin Score for Intraocular Lymphoma Diagnosis is a useful probability score for the diagnosis of PVRL that is based on a mathematical formula integrating both IL-10 and IL-6 levels in vitreous or aqueous samples with high sensitivity and specificity (93% and 95%, respectively).7
Finally, VRL should be suspected when there is nonspecific posterior uveitis with better visual acuity than expected, minimal anterior chamber flare, dense vitritis, and absence of posterior synechiae or cystoid macular edema.
UVEAL MELANOMA
Uveal melanoma is the most common primary intraocular malignancy, with an annual incidence of five to seven cases per one million individuals in the United States. It predominantly affects White individuals and has a slightly higher prevalence in men. Risk factors include light iris color, ultraviolet light exposure, and a genetic predisposition, such as mutations in the GNAQ, GNA11, or BAP1 genes.8
Melanomas appear as hypovascular, dome-shaped masses on B-scan ultrasonography and are characterized by a centrally located vessel with a “central pattern” of blood flow. Uveal melanoma may present as a masquerade syndrome, mimicking conditions such as endogenous endophthalmitis, subretinal fluid in the macula with pigment epithelial detachments, and a thickened choroid resembling central serous chorioretinopathy, choroidal vortex vein varix, or chronic unilateral uveitis.8-11
LEUKEMIA
About 90% of patients with leukemia, especially those with the chronic form, experience ocular involvement.12 In cases of acute leukemia, ocular signs often correlate with disease relapse or progression. Malignant cells have been documented to infiltrate the uvea, optic nerve, cranial nerves, and periorbital tissues.12,13 Ocular manifestations are more commonly associated with anterior uveitis presenting with hypopyon, while secondary extramedullary location of acute myeloid leukemia can present as anterior uveitis with dust-like pigmented keratic precipitates, iris bombe, ischemic bilateral retinal vasculitis, and anterior synechiae with retinal detachment and a lesion in the subretinal space.2 Posterior segment involvement may result from direct ocular infiltration or indirect effects of systemic disease.2,12 Retinal hemorrhage, vitreous hemorrhage, vascular occlusion, and secondary infections may present as indirect sequelae of systemic malignant disease.2,12,13
METASTATIC TUMOR
Metastatic tumors are the most common intraocular malignancy with the majority originating from breast (40%) and lung (30%) cancers. Choroidal metastases predominate due to the rich vascular supply of the posterior uvea. Many cases of lung cancer with masquerading ocular metastases have been documented, presenting as anterior uveitis with pseudokeratic precipitates, secondary glaucoma with goniosynechiae and iris nodules, bullous retinal detachment, and diffuse retinal vasculitis with vitritis and optic disc edema.14,15 Furthermore, ocular metastasis of mammary carcinoma may mimic bilateral anterior uveitis with hypopyon, partial third nerve palsy, and eyelid margin thickening.16,17
Fewer cases of ocular metastasis from kidney carcinoma have been reported, masquerading as endogenous endophthalmitis, while eyelid and ocular metastases from gastrointestinal carcinoma may present as chalazion.18,19 Ocular metastases are most commonly found in the choroid (88%), iris (9%), and ciliary body (2%). Metastases typically appear yellow in color and can be solitary, multiple, unilateral, or bilateral. They can cause blurred vision (70%), flashes and floaters (12%), or may be asymptomatic.18,19
TIME IS OF THE ESSENCE
The primary diagnostic method for masquerade syndromes remains the cytological and histological analysis of a vitreous biopsy via pars plana vitrectomy.5,15,20 Further research into molecular diagnostics and targeted treatments is essential to enhance prognosis and quality of life for affected patients.
1. Carbonell D, Mahajan S, Chee SP, et al. Consensus recommendations for the diagnosis of vitreoretinal lymphoma. Ocul Immunol Inflamm. 2021;29(3):507-520.
2. Paez-Escamilla M, Caplash S, Kalra G, et al. Challenges in posterior uveitis—tips and tricks for the retina specialist. J Ophthalmic Inflamm Infect. 2023;13(1):35.
3. Rothova A, Groen F, ten Berge JCEM, Lubbers SM, Vingerling JR, Thiadens AAHJ. Causes and clinical manifestations of masquerade syndromes in intraocular inflammatory diseases. Retina. 2021;41(11):2318-2324.
4. Kraan W, Horlings HM, Van Keimpema M, et al. High prevalence of oncogenic MYD88 and CD79B mutations in diffuse large B-cell lymphomas presenting at immune-privileged sites. Blood Cancer J. 2013;3(9):e139.
5. Pe’er J, Hochberg FH, Foster CS. Clinical review: treatment of vitreoretinal lymphoma. Ocul Immunol Inflamm. 2009;17(5):299-306.
6. Carbonell D, Mahajan S, Chee SP, et al. Study group for vitreoretinal lymphoma diagnostics. consensus recommendations for the diagnosis of vitreoretinal lymphoma. Ocul Immunol Inflamm. 2021;29(3):507-520.
7. Soussain C, Malaise D, Cassoux N. Primary vitreoretinal lymphoma: a diagnostic and management challenge. Blood. 2021;138(17):1519-1534.
8. Sara Simões Macedo, Mónica Teixeira, Andreia Correia, António Novais, Cátia Cabral. Ocular melanoma presenting as masquerade syndrome. Eur J Case Rep Intern Med. 2019;6(7):001118.
9. Feng L, Zhu J, Gao T, Li B, Yang Y. Uveal melanoma in the peripheral choroid masquerading as chronic uveitis. Optometry and Vision Science. 2014;91(9):e222-225.
10. Gillan SN, Roxburgh STD. Vortex varix manifesting as choroidal melanoma. BMJ. 2013;346(jun25 3):f3947-f3947.
11. Negretti GS, Kalafatis NE, Shields JA, Shields CL. Choroidal melanoma masquerading as central serous chorioretinopathy. Ophthalmol Retina. 2023;7(2):171-177.
12. Eze B, Ibegbulam G, Ocheni S. Ophthalmic manifestations of leukemia in a tertiary hospital population of adult Nigerian Africans. Middle East Afr J Ophthalmol. 2010;17(4):325.
13. Reddy SC, Jackson N, Menon BS. Ocular involvement in leukemia – a study of 288 cases. Ophthalmologica. 2003;217(6):441-445.
14. Kim KW, Kusuhara S, Tachihara M, Mimura C, Matsumiya W, Nakamura M. A case of panuveitis and retinal vasculitis associated with pembrolizumab therapy for metastatic lung cancer. Am J Ophthalmol Case Rep. 2021;22:101072.
15. Konno S, Yuzawa S, Kinouchi R. A case of masquerade syndrome caused by metastatic iris tumor diagnosed by a high CEA level in the aqueous humor and iris biopsy. Diagn Pathol. 2024;19(1):128.
16. Amer NM, Bashir G, Ogedegbe A, Saeed I. Metastatic breast cancer: an unusual cause of diplopia. J Cancer Metastasis Treat. 2016;2(3):123.
17. Annamalai R, Delampady P, Biswas J. Bilateral hypopyon uveitis in carcinoma of breast: Masquerade syndrome and review of literature. Indian J Ophthalmol Case Rep. 2023;3(4):1147-1150.
18. Rebollo NP, Yeaney GA, Hwang CJ, Perry JD. Metastatic gastric carcinoma to the eyelids masquerading as a chalazion: a case report. Am J Ophthalmol Case Rep. 2023;29:101814.
19. Ge JY, Loo YH, Chan ASY. Metastatic renal cell carcinoma masquerading as an endogenous endophthalmitis: role of vitreous cytology. Asia Pac J Ophthalmol (Phila). 2024;13(2):100055.
20. Coupland SE, Bechrakis NE, Anastassiou G, et al. Evaluation of vitrectomy specimens and chorioretinal biopsies in the diagnosis of primary intraocular lymphoma in patients with Masquerade syndrome. Graefes Arch Clin Exp Ophthalmol. 2003;241(10):860-870.














_1773249222.png?auto=compress,format&w=75)












