Following patients in real-world clinical practice is markedly different than clinical trial settings. Aside from the fact that the absence of strict follow-up protocols inherently alters when and how often patients are seen in the clinic, other considerations, such as how far a patient lives from the office, tend to take on greater prominence. As well, other medical concerns may complicate the risk-benefit analysis of offering treatment. Below is an example case from my practice that demonstrates one patient’s experience with geographic atrophy (GA), showing how real-world factors figure into the treatment algorithm.
Case Presentation
In April of 2022, a 71-year-old woman presented to my clinic with a history of GA OD. She was (and is) a current smoker who lives alone 4.5 hours away from my office. She reported that her daughter and granddaughter live closer to my office; nevertheless, the need to travel to my clinic would be a complicating factor. Relevant findings at the time of the exam were a history of fibrotic scar and neovascular AMD (nAMD) OS that has never been treated. The presenting BCVA was 20/40 OD and CF OS. Fundus autofluorescence (FAF) at the time of the exam showed multifocal lesions surrounding the fovea (Figure 1).
The patient was next seen in my office in October 2022, and she was examined a few times more before undergoing cataract surgery OU in July 2023. When she came to see me in August of 2023, FAF photos showed enlargement of the atrophic area (Figure 2) compared to April 2022. OCT captured during this visit demonstrated hypertransmission defects corresponding with the GA lesions (Figure 3). BCVA was 20/30 OD, CF OS. We discussed starting complement inhibition therapy at this visit, and we started treating her right eye with avacincaptad pegol (Izervay; Astellas) in November 2023.

Figure 2. FAF in August of 2023 showed progression of the GA lesions compared to the initial visit in April 2022 (as seen in Figure 1).
During a January 2024 visit, after 2 monthly injections of avancincaptad pegol, the patient complained of worsening vision OD. On OCT, she was found to have new cystoid macular edema, subretinal fluid, and subretinal hyperreflective material (Figure 4). She was diagnosed with new onset wet AMD and was treated with bevacizumab (Avastin). BCVA at this visit was 20/40. She returned a month later for follow-up, at which time the anatomy had improved, but BCVA was still 20/40 (Figure 5). A second bevacizumab injection was administered.
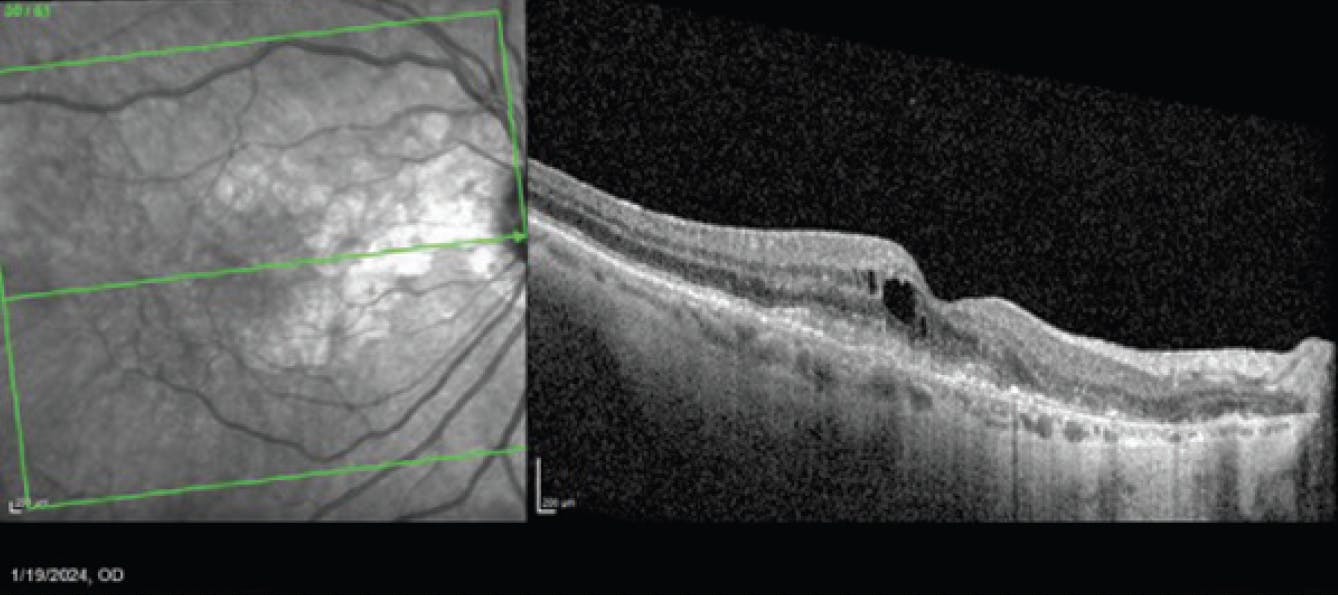
Figure 4. In January 2024, OCT (right) showed cystoid macular edema, subretinal fluid, and subretinal hyperreflective material consistent with wet AMD.
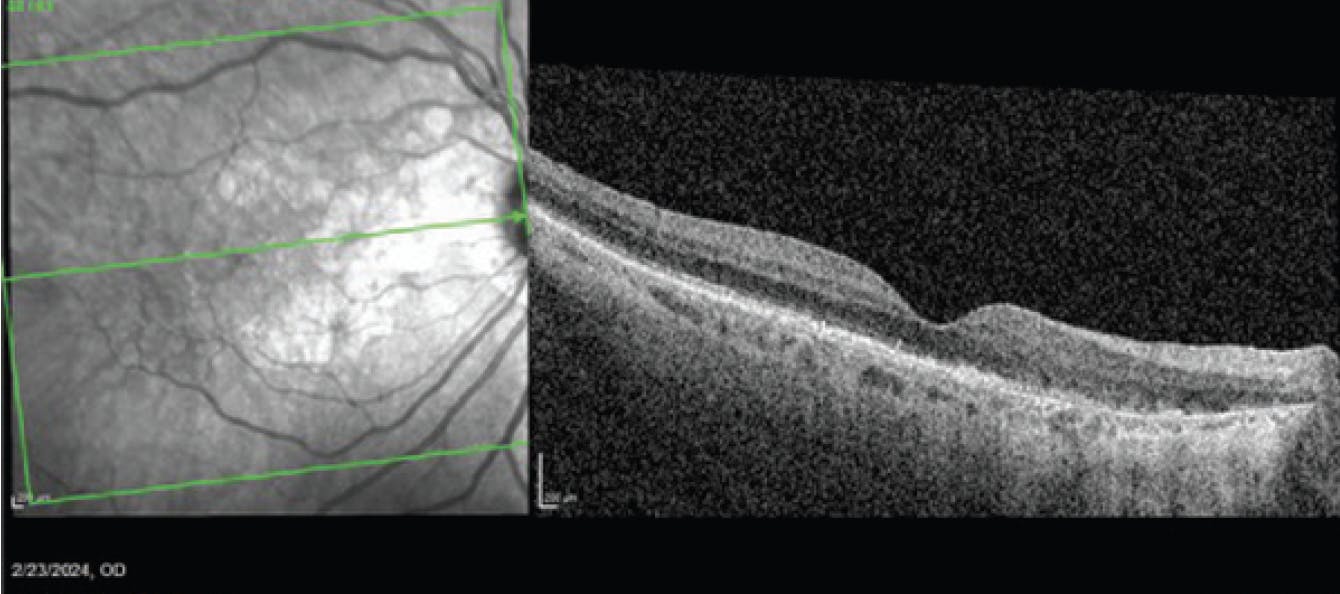
Figure 5. OCT of the patient in February 2024, 1 month after injection of an anti-VEGF agent to treat the wet AMD.
Additional Follow-Up and Long-Term Prognosis
This patient was subsequently seen a month later; BCVA at that time was 20/40. At this point, treatment for the GA was redosed with avacincaptad pegol, as the wet AMD appeared to be stable. Currently, the patient is treated on alternating months with bevacizumab and avacincaptad pegol. BCVA at the latest follow-up had improved to 20/30. Because complement inhibition therapy is intended to slow down the rate of GA progression (rather than restore vision), the goal of treatment with this patient is to preserve the remaining vision for as long as possible. This case highlights managing both wet AMD and GA in a monocular patient, and fortunately, for this patient, she has had a result thus far.







