Retinal vascular disorders in neonates, particularly oxygen-associated retinopathy (OAR) and retinopathy of prematurity (ROP), represent significant causes of preventable childhood blindness worldwide.1 While these conditions share some pathological features, they differ fundamentally in their etiology, clinical presentation, and management approaches.2 This article provides a comprehensive comparison of OAR and ROP based on current evidence, with particular emphasis on their distinctive clinical features and management strategies.
CLINICAL AND ANGIOGRAPHIC DIFFERENTIATION
OAR and ROP demonstrate distinct clinical presentations that enable differentiation through careful examination.3 OAR typically manifests with dichotomous branching of the retinal vessels and prominent peripapillary involvement, reflecting its pathogenesis from oxygen toxicity in both preterm and full-term infants; in contrast, ROP exhibits the characteristic menorah pattern of vascular growth, consistent with its developmental origin in premature infants with incomplete retinal vascularization (Figure 1).4,5
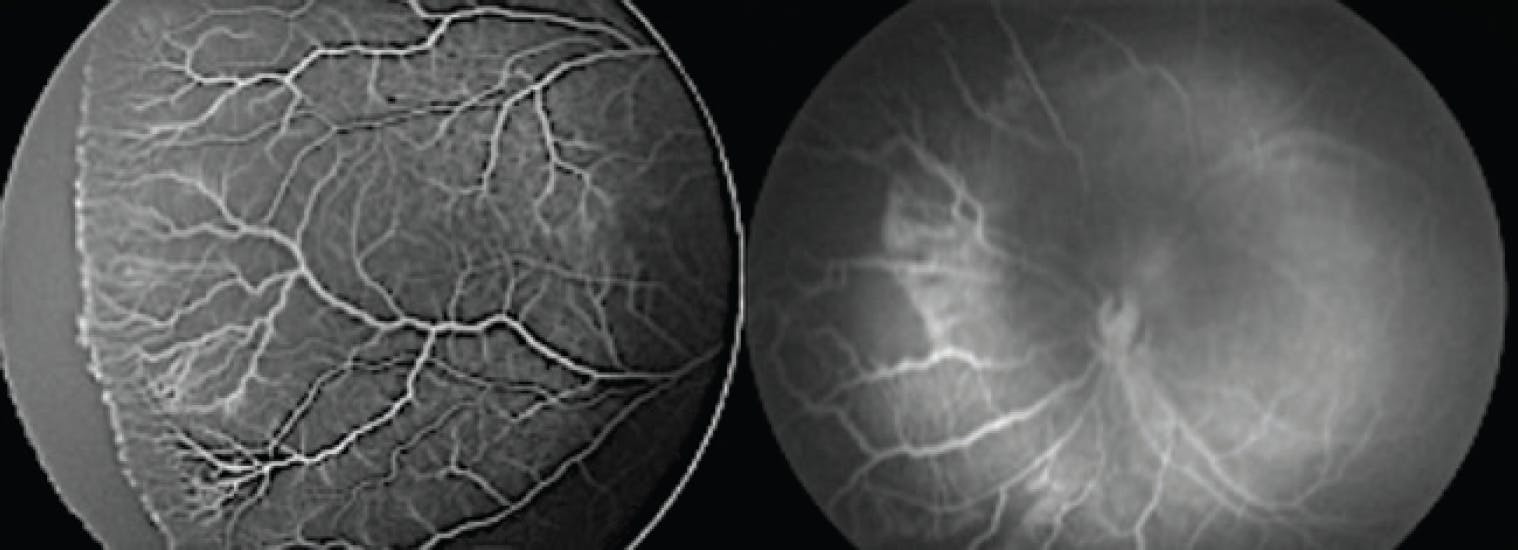
Figure 1. Vascular patterns in OAR and ROP. OAR shows dichotomous branching with peripapillary involvement, while ROP exhibits the classic menorah pattern of abnormal vascular growth.
Fluorescein angiography sheds light on further diagnostic differences between OAR and ROP.6 OAR demonstrates delayed arterial filling and patchy choroidal perfusion patterns, while ROP shows a clear demarcation between the vascular and avascular retina.7 These angiographic features not only aid in diagnosis, but also provide prognostic information regarding disease progression.8
DIVERGENT EARLY-STAGE MANAGEMENT
The most critical distinction between OAR and ROP lies in their differential response to early intervention.9 In OAR cases identified during the vaso-obliteration phase, carefully titrated oxygen therapy at 88% to 95% FiO₂ concentration can induce disease regression (Figure 2).10 This therapeutic window promotes vascular reperfusion and facilitates physiological growth of previously obliterated vessels.11
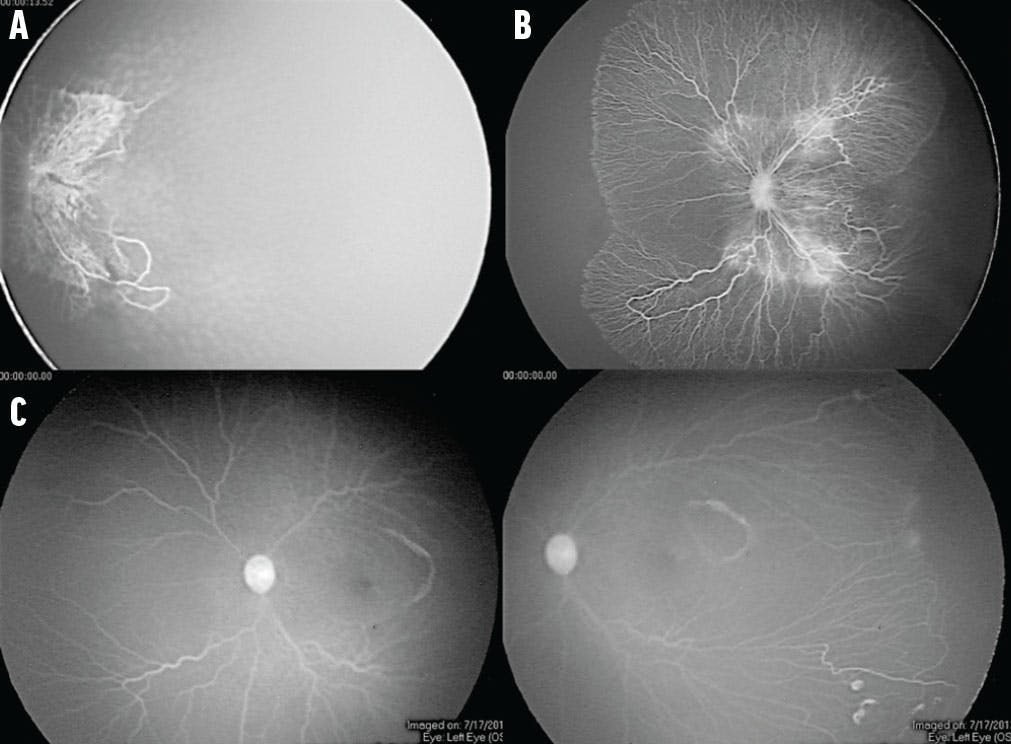
Figure 2. In OAR, carefully titrated oxygen therapy during the vaso-obliteration phase can promote regression and reperfusion of retinal vessels. A patient with aggressive vaso-obliteration before treatment (A) and 3 months (B) and 6 months (C) after regulated oxygen therapy has vascular reperfusion and physiological growth.
In contrast, early-stage ROP typically requires only careful observation, as oxygen modulation does not provide therapeutic benefit in this condition.12 This fundamental difference underscores the importance of accurate differentiation between these entities.13
ADVANCED DISEASE MANAGEMENT
If either condition progresses to neovascularization, their management converges, requiring antiangiogenic therapy with an anti-VEGF agent (Figure 3) or laser photocoagulation.6 For retinal detachment (stages 4 and 5), vitrectomy becomes necessary regardless of etiology.14 Stage 5 disease represents the most common indication for surgical intervention, although outcomes remain suboptimal.15

Figure 3. Before treatment with anti-VEGF therapy (A), capillary closure, ischemia, and aggressive neovascularization are obvious. Two weeks after anti-VEGF therapy (B), regression of neovascular changes is evident.
REHABILITATION AND GLOBAL HEALTH PERSPECTIVES
Postoperative rehabilitation is crucial for children with advanced disease.16 Translucent prostheses offer particular benefits for OAR patients by preserving residual light perception while maintaining orbital growth (Figure 4).
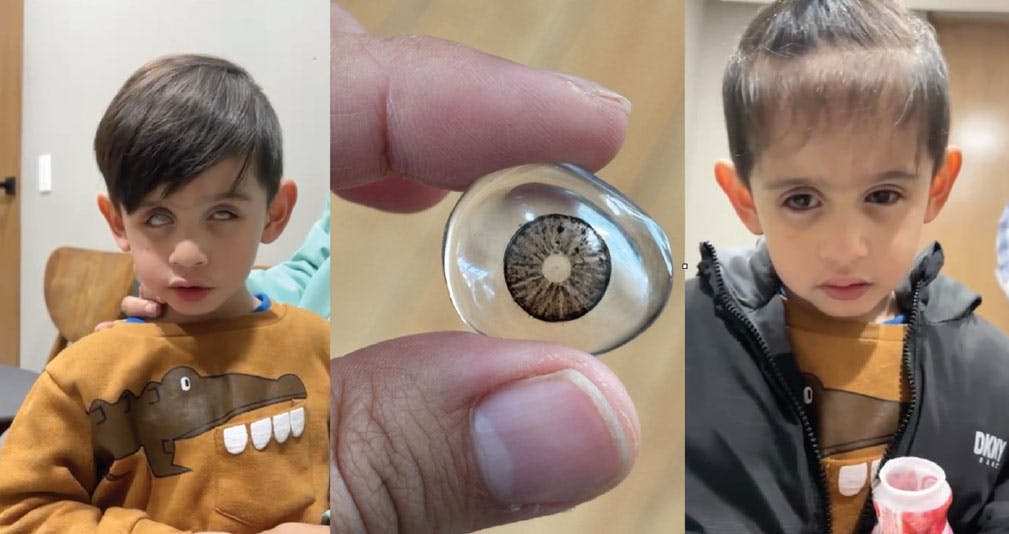
Figure 4. This child with stage 5 OAR is using a translucent prosthesis, which preserves residual light perception while supporting orbital growth during the postoperative rehabilitation.
The disproportionate burden of these conditions on children and families in low-income countries highlights health care disparities and the need for improved access to basic neonatal ophthalmic care.17
FIRST, DIFFERENTIATE
Accurate differentiation between OAR and ROP is essential for proper management and care.18 The unique responsiveness of OAR to oxygen modulation represents a critical window for vision preservation, while advanced cases of both conditions require similar interventions. Global efforts should focus more on implementing preventive strategies and improving access to care.
1. Smith LEH. Pathogenesis of retinopathy of prematurity. Semin Neonatol. 2003;8(6):469-473.
2. Hellström A, Smith LEH, Dammann O. Retinopathy of prematurity. Lancet. 2013;382(9902):1445-1457.
3. Flynn JT, Bancalari E, Bachynski BN, et al. Fluorescein angiography in retinopathy of prematurity. Ophthalmology. 1987;94(6):620-629.
4. Ashton N. Oxygen and the growth and development of retinal vessels. Am J Ophthalmol. 1966;62(3):412-435.
5. International Committee for the Classification of Retinopathy of Prematurity. The International Classification of Retinopathy of Prematurity revisited. Arch Ophthalmol. 2005;123(7):991-999.
6. Mintz-Hittner HA, Kennedy KA, Chuang AZ. Efficacy of intravitreal bevacizumab for stage 3+ retinopathy of prematurity. N Engl J Med. 2011;364(7):603-615.
7. Palmer EA, Flynn JT, Hardy RJ, et al. Incidence and early course of retinopathy of prematurity. Ophthalmology. 1991;98(11):1628-1640.
8. Early Treatment for Retinopathy of Prematurity Cooperative Group. Revised indications for the treatment of retinopathy of prematurity. JAMA Ophthalmol. 2003;121(12):1684-1694.
9. Chen J, Smith LEH. Retinopathy of prematurity. Angiogenesis. 2007;10(2):133-140.
10. Hartnett ME, Lane RH. Effects of oxygen on the development and severity of retinopathy of prematurity. J AAPOS. 2013;17(3):229-34.
11. Hartnett ME, Penn JS. Mechanisms and management of retinopathy of prematurity. N Engl J Med. 2012;367(26):2515-2526.
12. Gilbert C, Fielder A, Gordillo L, et al. Characteristics of infants with severe retinopathy of prematurity in countries with low, moderate, and high levels of development: implications for screening programs. Pediatrics. 2005;115(5):e518-25.
13. Quinn GE, Ying G, Daniel E, et al. Validity of a telemedicine system for the evaluation of acute-phase retinopathy of prematurity. JAMA Ophthalmol. 2014;132(10):1178-1184.
14. Trese MT. Surgical results of stage V retrolental fibroplasia and timing of surgical repair. Ophthalmology. 1984;91(5):461-466.
15. Sen P, Jain S, Bhende P. Stage 5 retinopathy of prematurity: an update. Taiwan J Ophthalmol. 2018;8(4):205-215.
16. Khalil H, de Moel-Mandel C, Verma D, et al. Characteristics of quality improvement projects in health services: a systematic scoping review. J Evid Based Med. 2025;18(1):e12670.
17. Blencowe H, Lawn JE, Vazquez T, Fielder A, Gilbert C. Preterm-associated visual impairment and estimates of retinopathy of prematurity at regional and global levels for 2010. Pediatr Res. 2013;74(1):35-49.
18. Darlow BA, Vento M, Beltempo M, et al. Variations in oxygen saturation targeting, and retinopathy of prematurity screening and treatment criteria in neonatal intensive care units. Neonatology. 2018;114(4):323-331.









_1773249222.png?auto=compress,format&w=75)












